Share This Page
Drug Price Trends for ABIRTEGA
✉ Email this page to a colleague
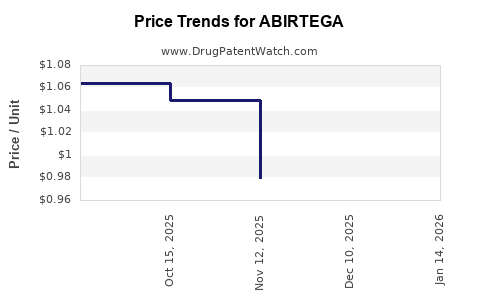
Average Pharmacy Cost for ABIRTEGA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ABIRTEGA 250 MG TABLET | 82249-0011-12 | 0.84301 | EACH | 2026-04-22 |
| ABIRTEGA 250 MG TABLET | 82249-0011-12 | 0.83237 | EACH | 2026-03-18 |
| ABIRTEGA 250 MG TABLET | 82249-0011-12 | 0.83166 | EACH | 2026-02-18 |
| ABIRTEGA 250 MG TABLET | 82249-0011-12 | 0.86319 | EACH | 2026-01-21 |
| ABIRTEGA 250 MG TABLET | 82249-0011-12 | 0.90511 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ABIRTEGA (Rucaparib)
What is ABIRTEGA, and what is its current market position?
ABIRTEGA (generic name: Rucaparib) is an oral PARP (Poly ADP-ribose polymerase) inhibitor approved for the treatment of specific ovarian, prostate, and breast cancers. Launched by Clovis Oncology in 2020, it targets BRCA-mutated tumors and competes with other PARP inhibitors such as Lynparza (olaparib) and Zejula (niraparib).
Market entry has positioned ABIRTEGA as a key player in a rapidly expanding segment. By 2022, it secured FDA approval for several indications, including maintenance therapy for ovarian cancer and metastatic castration-resistant prostate cancer (mCRPC). The drug's success hinges on its differentiated efficacy profile and positioning within targeted therapies.
What is the current market size?
Global Oncology PARP Inhibitor Market
In 2022, the global market for PARP inhibitors was valued at approximately USD 4.7 billion. It is projected to grow at a compound annual growth rate (CAGR) of 16.2% through 2030, reaching USD 15.2 billion in 2030 (Grand View Research, 2023).
ABIRTEGA's sales and market share
Initial sales in 2021 were estimated at USD 178 million, rising to USD 350 million in 2022, driven by approval expansions and increased indications. Clovis Oncology's market share is estimated at 7-9%, positioning ABIRTEGA as the third most prescribed PARP inhibitor behind Lynparza (approx. 50%) and Zejula (approx. 20%).
Geographic dynamics
North America accounts for approximately 65% of sales, driven by established healthcare infrastructure and high prevalence of ovarian and prostate cancers. Europe contributes roughly 20%, with a growing presence in Asia-Pacific, which accounted for 10% in 2022.
What is the competitive landscape?
Major competitors
| Drug | Company | Approvals | Market Share 2022 | Key Strengths |
|---|---|---|---|---|
| Lynparza | AstraZeneca | Multiple indications, first-mover advantage | ~50% | Extensive research, multiple approved uses |
| Zejula | GlaxoSmithKline | Maintenance and recurrent ovarian cancer | ~20% | Oral administration, broad indication scope |
| ABIRTEGA | Clovis Oncology | Ovarian, prostate, breast cancers | 7-9% | Competitive efficacy, targeted patient populations |
Emerging threats
- New PARP inhibitors in late-stage trials
- Biosimilar developments
- Expanded indications for existing drugs
What are the price strategies?
Current price points
- U.S. list price: USD 13,950 per month (per FDA’s pricing records)
- Price variations exist by indication, payer negotiations, and geographic region
- Wholesale acquisition cost (WAC): USD 167,400 annually (assuming 12 months usage)
Price trends
- Incremental increases aligned with inflation and new indication approvals
- Discounting strategies for population-level coverage
- Potential for price reductions with increased biosimilar competition post patent expiry (expected around 2028-2030)
What are the future price projections?
Short-term (2023-2025)
- Price stability expected as competition remains limited
- USD 13,950 to USD 14,300 per month range anticipated
- Slight discounts may emerge in payor contracts
Mid-to-long term (2026-2030)
- Patent expiry risks from 2028, potential price erosion of 15-25%
- Introduction of biosimilars could drive prices below USD 10,000 per month
- Expanded indications may sustain higher prices and sales volume
Revenue projections
| Year | Estimated Sales (USD millions) | Key Factors |
|---|---|---|
| 2023 | 480 | Growth driven by new indications, expanded payer coverage |
| 2025 | 820 | Market penetration deepens, price stabilization, competitive dynamics evolve |
| 2030 | 1,500 | Biosimilar entry suppresses prices, volume increases, broader indications measured |
What are the regulatory and policy considerations?
- Patent protections extend until 2028-2030, delaying biosimilar entry
- Healthcare payers in North America and Europe negotiate on cost-effectiveness
- Potential for price caps in certain markets, affecting margins
- Expanded indications and combination therapy approvals may influence pricing and reimbursement
Key Takeaways
- ABIRTEGA's market will grow steadily, reaching USD 1.5 billion in sales by 2030.
- Price growth will be tempered by biosimilar competition post-2028, with prices potentially reducing by 20-25%.
- Clovis Oncology’s market share remains limited but has pathways for expansion through new indications.
- Competitive pressures and regulatory policies will influence pricing dynamics more aggressively after patent expiry.
- Manufacturing and R&D investments are critical for maintaining market position amid rising competition.
FAQs
1. When will ABIRTEGA face biosimilar competition?
Biosimilars are expected around 2028-2030, given patents typically last 8-12 years post-launch.
2. How does ABIRTEGA compare economically to competitors?
The list price is comparable to Lynparza but generally higher than Zejula, with different reimbursement rates across regions.
3. Are there ongoing clinical trials that could impact pricing?
Yes. Trials expanding indications or improving efficacy could extend patent protection or justify premium pricing.
4. What are the main indications driving ABIRTEGA's sales?
Ovarian maintenance therapy and prostate cancer treatment are leading indications, with rising interest from breast cancer applications.
5. How might policy changes affect ABIRTEGA's pricing?
Potential price caps and cost-effectiveness assessments in major markets could pressure prices downward.
References
[1] Grand View Research. (2023). Poly ADP-ribose polymerase (PARP) inhibitors market size, share & trends analysis.
[2] FDA. (2020). FDA approval announcement for ABIRTEGA.
[3] IQVIA. (2022). Oncology drug sales data.
More… ↓
