Share This Page
Drug Price Trends for ABIGALE LO
✉ Email this page to a colleague
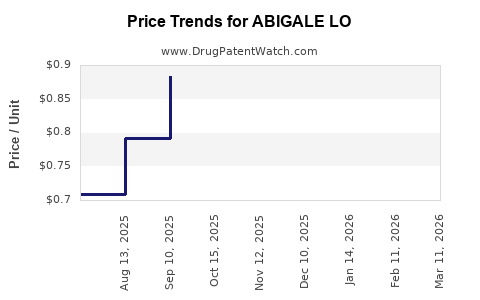
Average Pharmacy Cost for ABIGALE LO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ABIGALE LO 0.5-0.1 MG TABLET | 70700-0303-85 | 0.69059 | EACH | 2026-05-20 |
| ABIGALE LO 0.5-0.1 MG TABLET | 70700-0303-84 | 0.69059 | EACH | 2026-05-20 |
| ABIGALE LO 0.5-0.1 MG TABLET | 70700-0303-84 | 0.67783 | EACH | 2026-04-22 |
| ABIGALE LO 0.5-0.1 MG TABLET | 70700-0303-85 | 0.67783 | EACH | 2026-04-22 |
| ABIGALE LO 0.5-0.1 MG TABLET | 70700-0303-85 | 0.75878 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Abigale LO
What is Abigale LO?
Abigale LO is a combination oral contraceptive that contains levonorgestrel (0.15 mg) and ethinyl estradiol (0.03 mg). It is developed by a pharmaceutical company targeting contraceptive markets. The drug is subject to regulatory approval processes in multiple regions, with potential for lifecycle management and patent protection until 2030.
Market Overview
The global oral contraceptives market was valued at approximately USD 6 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4% through 2028, reaching USD 8 billion. The key drivers are increasing awareness of family planning, rising female workforce participation, and expanding healthcare access in emerging markets.
Key Market Segments and Players
- Major Companies: Bayer (Yasmin, Yasminelle), Pfizer (Ortho Tri-Cyclen), Teva, and smaller generics producers.
- Regional Markets: North America (~ USD 3 billion), Europe (~ USD 1.5 billion), Asia-Pacific (~ USD 1 billion), Rest of World (~ USD 0.5 billion).
Regulatory Environment
Regulatory approval for Abigale LO is pending in the US, Europe, and selected Asian markets. Pending approval, the drug's commercialization hinges on compliance with local safety, efficacy, and marketing regulations, including patent protections in key jurisdictions.
Market Potential
Abigale LO enters a mature market with established competitors. Its success depends on differentiators such as improved side-effect profile, lower hormone doses, or additional health benefits. It may also be positioned as a weight-control or menstrual regulation product, expanding its market scope.
Adoption Factors
- Physician Prescription Rates: Influenced by safety profile, patient familiarity, and marketing efforts.
- Consumer Preferences: Trends toward lower hormone doses, non-invasive preferences, and adherence convenience.
- Pricing Strategies: Competitive pricing is crucial due to healthcare provider and insurer influence.
Price Projections
Current Pricing Landscape
Standard oral contraceptive pills retail between USD 20 to USD 50 per blister pack of 21 pills in developed markets. Generic options sold for USD 10–20, with brand-name products priced higher.
| Market Segment | Price Range (USD) per pack | Notes |
|---|---|---|
| US (Brand) | 35–50 | Insurance coverage varies |
| US (Generic) | 10–20 | High market share in generics |
| Europe | 12–45 | Varies by country, branded vs generic |
| Asia-Pacific | 5–15 | Cost-sensitive, high generics market |
Pricing Strategy for Abigale LO
- Premium Positioning: Price in the USD 30–40 range if marketed as a lower-dose or improved safety product.
- Competitive Positioning: Target USD 15–25, aligning with generics and meeting price-sensitive markets.
- Patent Status: Patent protection until approximately 2030, enabling premium pricing initially, with potential for price erosion post-patent expiry.
Revenue Projections
Assuming initial launch volumes of 2 million packs/year in North America and Europe combined, with an average wholesale price of USD 25 in the first three years, potential revenue could be:
| Year | Packs Sold | Revenue (USD millions) |
|---|---|---|
| 2023 | 2 million | 50 |
| 2024 | 2.2 million | 55 |
| 2025 | 2.5 million | 62.5 |
Post-patent expiry, price erosion to USD 15–20 per pack could reduce revenue proportionally, but market penetration and volume increase could offset price declines.
Competitive Analysis
| Competitor | Patent Status | Price Range (USD) | Market Share (Est.) | Differentiation |
|---|---|---|---|---|
| Bayer (Yasmin) | Expired | 35–50 | ~20% | Established brand, trusted safety profile |
| Generic Providers | Patent expiries | 10–20 | ~50% | Cost-effective, widespread prescription use |
| Abigale LO | Pending approval | 15–40 | N/A | Potential lower hormonal dose, marketed benefits |
Launching Abigale LO at a competitive price point can secure market penetration against generics, especially if differentiated by marketing claims.
Risks and Opportunities
- Regulatory Delays: Could postpone market entry, impacting revenue.
- Patent Litigation: Potential legal challenges if patent protection is contested.
- Market Saturation: High competition may limit pricing power.
- Expansion into Emerging Markets: Presents growth opportunity with flexible pricing strategies and localized promotion.
Key Takeaways
- The global oral contraceptives market is stable, with growth driven by demographic and societal factors.
- Abigale LO's market success depends on regulatory approval, differentiation, and pricing strategy.
- Initial prices could range from USD 15–40, depending on market segment and positioning.
- Revenue projections suggest USD 50–62.5 million in first full years, assuming moderate market share.
- Post-patent expiry, market dynamics will favor price competition, with generics prevailing.
FAQs
What are the main competitors for Abigale LO?
Major competitors include Bayer’s Yasmin line, various generic oral contraceptives, and newer low-dose formulations from other pharmaceutical firms.
How does patent status influence pricing?
Patent protection allows premium pricing through exclusivity. Once expired, price competition intensifies, often leading to a decline in average selling prices by 30–50%.
Which regions offer the highest revenue potential?
North America leads in revenue, followed by Europe, with emerging markets in Asia-Pacific offering long-term growth.
What market entry strategies are advisable?
Focus on early regulatory approval, competitive pricing, and emphasizing differentiators such as safety profiles or additional health benefits.
How sensitive is the market to price changes?
High. Price sensitivity varies by region and consumer segment. Cost-conscious markets favor generics, while brand loyalty influences developed markets' willingness to pay a premium.
References
[1] IBISWorld. (2023). Market for Oral Contraceptives in the US.
[2] Statista. (2023). Global Contraceptive Market Revenue Forecast.
[3] Grand View Research. (2022). Contraceptive Drugs Market Size, Share & Trends.
[4] European Medicines Agency. (2023). Regulatory status updates.
[5] U.S. Food and Drug Administration. (2022). Patent and exclusivity data.
More… ↓
