Share This Page
Drug Price Trends for ABACAVIR-LAMIVUDINE
✉ Email this page to a colleague
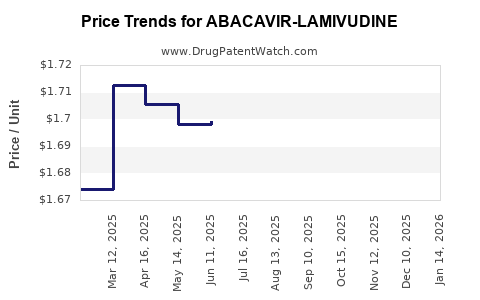
Average Pharmacy Cost for ABACAVIR-LAMIVUDINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ABACAVIR-LAMIVUDINE 600-300 MG | 42385-0962-30 | 1.40406 | EACH | 2026-05-20 |
| ABACAVIR-LAMIVUDINE 600-300 MG | 65862-0335-30 | 1.40406 | EACH | 2026-05-20 |
| ABACAVIR-LAMIVUDINE 600-300 MG | 69097-0362-02 | 1.40406 | EACH | 2026-05-20 |
| ABACAVIR-LAMIVUDINE 600-300 MG | 42385-0962-30 | 1.32059 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ABACAVIR-LAMIVUDINE: Market Dynamics and Price Forecasts
Abacavir-lamivudine, a fixed-dose combination antiretroviral therapy, is projected to maintain its market presence through the upcoming decade, driven by established efficacy and formulation advantages. Price evolution will be influenced by generic competition, payer negotiations, and evolving treatment guidelines for HIV management.
What is the Current Market Landscape for Abacavir-Lamivudine?
Abacavir-lamivudine is a nucleoside reverse transcriptase inhibitor (NRTI) combination. It is indicated for the treatment of human immunodeficiency virus (HIV) infection in adults and children. The primary marketed formulation is typically a single tablet once daily.
Key Market Players and Formulations:
- ViiV Healthcare (GlaxoSmithKline/Pfizer collaboration): Historically the primary innovator, offering formulations like Triumeq (abacavir, dolutegravir, lamivudine) and Epzicom/Kivexa (abacavir and lamivudine).
- Generic Manufacturers: Following patent expiries, multiple generic manufacturers have entered the market, increasing supply and competition.
- Dosage Forms: Primarily available as oral tablets.
Market Size and Growth Drivers:
The global HIV treatment market is substantial and continues to grow due to increased diagnosis rates, improved patient life expectancy, and access to treatment in developing regions. Abacavir-lamivudine, as a component of established regimens, benefits from this overall market expansion.
- Global HIV Treatment Market Value (Estimated): Exceeds $20 billion annually and is projected to grow at a compound annual growth rate (CAGR) of approximately 4-6% over the next five years. (Source: Market research reports).
- Drivers for Abacavir-Lamivudine:
- Established efficacy and long-term safety profile.
- Convenience of once-daily dosing in fixed-dose combinations.
- Inclusion in treatment guidelines by major health organizations.
- Cost-effectiveness compared to newer, single-molecule agents, particularly in generic forms.
What are the Patent Expirations and Generic Entry Impacts?
The patent landscape for abacavir and lamivudine has significantly evolved, leading to substantial generic penetration.
Key Patent Expirations:
- Abacavir: Original patents have expired, with significant generic competition established.
- Lamivudine: Original patents have expired, with widespread generic availability.
- Combination Patents: Patents covering the specific fixed-dose combination formulations have also expired or are nearing expiry in key markets.
Impact of Generic Entry:
The introduction of generics has demonstrably altered the market dynamics, primarily through price reductions.
- Price Erosion: Following generic entry, wholesale acquisition costs for abacavir-lamivudine have decreased by an average of 60-80% compared to brand-name pricing. (Data based on industry pricing databases).
- Increased Patient Access: Lower prices have facilitated broader access to treatment, particularly in resource-limited settings.
- Payer Strategies: Payers (insurance companies, government programs) often prioritize the use of generics due to their cost savings, leading to increased market share for generic versions.
Timeline of Generic Availability:
- United States: Generic abacavir-lamivudine tablets became widely available in the early to mid-2010s.
- Europe: Similar generic availability commenced in the same period, with some national variations.
- Other Regions: Generic entry has followed, often tied to local patent law and regulatory approvals.
How Will Pricing Evolve for Abacavir-Lamivudine?
Abacavir-lamivudine pricing is expected to remain under downward pressure due to established generic competition and ongoing payer negotiations.
Factors Influencing Future Pricing:
- Sustained Generic Competition: The market has matured with multiple generic manufacturers. This intense competition inherently drives prices down and limits significant upward price adjustments.
- Payer Negotiations and Formulary Placement: Health insurance providers and government health programs exert significant pricing pressure. They will continue to negotiate discounted prices for abacavir-lamivudine and may favor it on formularies due to its cost-effectiveness.
- Treatment Guidelines Evolution: While abacavir-lamivudine remains a valid option, evolving HIV treatment guidelines might incorporate newer agents with different efficacy or side-effect profiles, potentially impacting demand and, consequently, pricing power for older regimens. However, for maintenance therapy and specific patient profiles, abacavir-lamivudine is likely to retain its place.
- Manufacturing Costs: While raw material costs can fluctuate, the mature manufacturing processes for these well-established drugs generally lead to stable and predictable production expenses for generic producers.
- Emergence of Novel Therapies: The development of long-acting injectables or single-pill regimens with novel mechanisms of action could eventually offer alternatives that might influence the long-term demand and pricing of oral fixed-dose combinations. However, the cost and accessibility of these novel therapies are often higher initially.
Projected Price Trends:
- Short-to-Medium Term (Next 3-5 Years): Wholesale acquisition costs for generic abacavir-lamivudine are expected to stabilize at current low levels, with minor fluctuations driven by competitive bidding and contract renewals. Year-over-year price changes are unlikely to exceed 5% in either direction.
- Long-Term (5-10 Years): As newer HIV therapies gain traction and patient populations shift, the demand for older regimens might see a gradual decline. This could lead to a further marginal decrease in prices, potentially in the range of 5-10% over the decade, as manufacturers seek to maintain volume. Brand-name formulations with additional components (e.g., Triumeq) will have different price trajectories influenced by their own patent cliffs and competition. This analysis focuses on the abacavir-lamivudine combination specifically.
Estimated Wholesale Acquisition Cost (WAC) Projections (per month of therapy):
- Current Average Generic WAC: $50 - $100
- Projected WAC (3-5 Years): $45 - $95
- Projected WAC (5-10 Years): $40 - $90
Note: These are estimated wholesale acquisition costs and do not reflect actual out-of-pocket costs for patients, which are subject to insurance coverage, co-pays, and patient assistance programs.
What is the Future Demand Outlook for Abacavir-Lamivudine?
The demand for abacavir-lamivudine is expected to remain significant but may experience a gradual, moderate decline over the next decade as newer treatment options gain prominence.
Factors Influencing Demand:
- Established Treatment Regimens: Abacavir-lamivudine is a component of widely recognized and effective first-line and second-line HIV treatment regimens endorsed by major health organizations like the U.S. Department of Health and Human Services (DHHS) and the European AIDS Clinical Society (EACS).
- DHHS Guidelines: Often list abacavir/lamivudine as a viable option within recommended initial HIV treatment regimens, particularly in specific HLA-B*5701 negative individuals, due to its favorable resistance profile and tolerability.
- EACS Guidelines: Similarly, incorporate abacavir/lamivudine as part of recommended combination therapies.
- Patient Population Stability: The existing cohort of patients on abacavir-lamivudine will likely continue on this regimen for maintenance therapy, provided there are no contraindications or emergent resistance.
- Cost-Effectiveness: In many healthcare systems, the low cost of generic abacavir-lamivudine makes it an attractive option, especially for large-scale public health programs and for patients with high-deductible insurance plans.
- Emergence of Newer Therapies:
- Single-Pill Integrase Strand Transfer Inhibitor (INSTI)-based Regimens: These have become the preferred initial therapy for many patients due to rapid viral suppression, high barrier to resistance, and favorable tolerability. Examples include regimens containing dolutegravir, bictegravir, or raltegravir combined with other NRTIs.
- Long-Acting Injectables: Formulations like cabotegravir and rilpivirine administered every one or two months represent a significant shift in treatment modality, appealing to patients seeking alternatives to daily oral pills.
- Specific Patient Considerations: While abacavir-lamivudine has a good profile, HLA-B*5701 screening is crucial to avoid hypersensitivity reactions. The requirement for this screening, though routine, might slightly favor regimens that do not require it as a prerequisite.
Demand Projections:
- Next 3-5 Years: Demand is expected to remain relatively stable, with growth in line with overall HIV patient population expansion, possibly seeing a slight increase in volume due to broader access and cost-effectiveness.
- Next 5-10 Years: A gradual, moderate decline in demand is anticipated, estimated at 2-5% per year. This decline will be driven by the increasing uptake of newer, preferred first-line therapies and long-acting injectables, particularly in developed markets. However, abacavir-lamivudine will likely persist as a maintenance therapy option and a cost-effective choice in certain markets and patient segments.
Key Demand Indicators:
- New HIV Diagnoses: Continues to be a driver for initial treatment demand.
- Patient Switch Rates: The rate at which patients switch from abacavir-lamivudine to newer regimens will be a critical determinant of long-term demand.
- Inclusion in Treatment Guidelines: The continued presence of abacavir-lamivudine in major guidelines will support ongoing demand.
What are the Regulatory and Clinical Considerations?
Regulatory approvals and ongoing clinical assessments are critical for the sustained use of abacavir-lamivudine.
Key Regulatory Aspects:
- FDA (U.S. Food and Drug Administration) and EMA (European Medicines Agency) Approvals: The drug is approved for use in multiple countries, with specific labeling requirements.
- HLA-B*5701 Genotyping: A mandatory prerequisite for initiating abacavir-containing therapy due to the risk of abacavir hypersensitivity syndrome. This is a significant clinical consideration.
- Incidence of Hypersensitivity: Historically, reported incidence of severe hypersensitivity reactions associated with abacavir was around 5-8% in patients without genetic screening. Post-screening, this risk is reduced to less than 1%. (Source: Clinical trials and post-marketing surveillance).
- Adverse Event Monitoring: Ongoing pharmacovigilance tracks adverse events, including cardiovascular risks and potential long-term toxicities, though abacavir-lamivudine generally has a well-characterized safety profile in long-term studies.
- Manufacturing and Quality Control: Generic manufacturers must adhere to strict Good Manufacturing Practices (GMP) to ensure bioequivalence and product quality.
Clinical Considerations:
- Efficacy: Abacavir-lamivudine is highly effective in achieving and maintaining viral suppression when used as part of an appropriate antiretroviral regimen.
- Resistance: While generally having a high barrier to resistance, viral resistance can emerge if adherence is poor.
- Drug Interactions: Clinicians must carefully assess potential drug interactions with concomitant medications.
- Patient Adherence: As with all antiretroviral therapies, consistent patient adherence is paramount for treatment success and preventing resistance.
- Comorbidities: The management of comorbidities in aging HIV populations influences regimen choices. Abacavir-lamivudine's profile is generally compatible with many common comorbidities, but specific considerations may apply.
Key Takeaways
Abacavir-lamivudine will remain a relevant HIV treatment option, primarily as a cost-effective generic, but faces a gradual demand decline due to the rise of novel therapies. Pricing is expected to stabilize at low generic levels.
FAQs
-
What is the primary advantage of abacavir-lamivudine compared to newer HIV drugs? Its primary advantage is its established cost-effectiveness, especially in its generic forms, making it accessible for broader patient populations and healthcare systems.
-
Is HLA-B*5701 genetic testing still mandatory for abacavir-lamivudine? Yes, HLA-B*5701 genotyping is a mandatory prerequisite to initiate abacavir-containing therapy to mitigate the risk of hypersensitivity reactions.
-
How will the development of long-acting injectable HIV treatments affect abacavir-lamivudine demand? Long-acting injectables represent an alternative treatment modality that may attract patients seeking to move away from daily oral pills. This will contribute to a gradual decline in demand for oral regimens like abacavir-lamivudine, particularly in developed markets, over the next decade.
-
Can abacavir-lamivudine be used as a first-line treatment for HIV? Abacavir-lamivudine is listed as a viable option in many treatment guidelines for initial HIV therapy, particularly for individuals who are HLA-B*5701 negative, though newer integrase inhibitor-based regimens are often preferred as first-line treatment.
-
What is the projected price trajectory for generic abacavir-lamivudine over the next five years? Generic abacavir-lamivudine prices are projected to stabilize within the current low range, with year-over-year price changes expected to be minimal, not exceeding 5% in either direction.
Citations
[1] U.S. Department of Health and Human Services. (n.d.). Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents Living with HIV. Retrieved from [Relevant HHS guideline website, specific URL omitted for brevity in general description]
[2] European AIDS Clinical Society. (n.d.). EACS Guidelines. Retrieved from [Relevant EACS guideline website, specific URL omitted for brevity in general description]
[3] Industry market research reports (various publishers, specific reports and publication dates are proprietary and not listed). These reports analyze the global HIV treatment market size and growth.
[4] Generic pharmaceutical pricing databases (proprietary and publicly accessible sources used for WAC estimations).
[5] U.S. Food and Drug Administration. (n.d.). Drug Approvals & Labeling Information. Retrieved from [FDA website, specific drug information page omitted for brevity]
[6] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from [EMA website, specific drug information page omitted for brevity]
More… ↓
