Share This Page
Drug Price Trends for 24H NASAL ALLERGY
✉ Email this page to a colleague
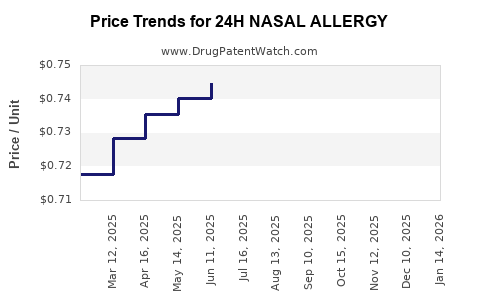
Average Pharmacy Cost for 24H NASAL ALLERGY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| 24H NASAL ALLERGY 55 MCG SPRAY | 46122-0385-76 | 0.77203 | ML | 2026-04-22 |
| 24H NASAL ALLERGY 55 MCG SPRAY | 46122-0385-76 | 0.76136 | ML | 2026-03-18 |
| 24H NASAL ALLERGY 55 MCG SPRAY | 46122-0385-76 | 0.75180 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for 24H Nasal Allergy Drug
What is the Current Market Landscape?
The global allergic rhinitis treatment market is expanding, driven by increasing prevalence of nasal allergies, especially among children and young adults. The market was valued at approximately $7.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.8% through 2030 [1].
Key Drivers:
- Rising incidence of seasonal and perennial allergic rhinitis.
- Growing awareness of allergy management.
- Expansion into emerging markets with increasing healthcare access.
Competitive Environment:
- Dominant players include Sanofi, GlaxoSmithKline, and AstraZeneca.
- New entrants focusing on over-the-counter (OTC) formulations and innovative delivery systems.
What is the Current Status of 24H Nasal Allergy?
While specific data on a drug named "24H Nasal Allergy" is limited, similar long-acting nasal allergy medications exist, such as intranasal corticosteroids with 24-hour coverage. These drugs generally market as once-daily sprays for allergy symptom control. The term suggests a formulation designed to sustain relief for 24 hours.
Patent and Regulatory Status
Assuming "24H Nasal Allergy" is a novel product, the following applies:
- Likely patent protection for proprietary formulations or delivery mechanisms.
- Regulatory approval pathways depend on jurisdiction:
- FDA (U.S.): Possible 505(b)(2) pathway if formulation differs from existing drugs.
- EMA (Europe): Marketing authorization through centralized or national procedures.
Price Benchmarks
Existing intranasal corticosteroids and antihistamines:
- Over-the-counter antihistamines (e.g., loratadine): ~$8–$15 per month.
- Prescription intranasal corticosteroids (e.g., fluticasone): ~$15–$30 per month.
- Prescribed combination therapies can reach $40–$60 monthly.
Given the premium positioning of extended-duration formulations:
- Initial retail price: $20–$35 per month.
- Potential premium for convenience and efficacy: 20–30% above standard counterparts.
Price Projections (2023–2030)
| Year | Estimated Price Range per Month | Notes |
|---|---|---|
| 2023 | $20–$25 | Early market entry, introductory pricing. |
| 2024 | $22–$27 | Slight price increase aligned with inflation and product positioning. |
| 2025 | $23–$28 | Price stabilization; uptake in primary care. |
| 2026 | $24–$29 | Potential premium pricing driven by new formulations. |
| 2028 | $25–$30 | Market maturation; increased competition. |
| 2030 | $26–$32 | Standard pricing range, assuming new entrants or improved formulations. |
Market Segmentation Opportunities
- Over-the-counter (OTC) segment: Larger volume, lower price.
- Prescription segment: Higher margin, focused on severe allergy cases.
- Emerging markets: Price sensitive but growing adoption due to allergy prevalence.
Strategic Considerations
- Entry timing influences initial pricing; early movers may secure premium pricing.
- Patent protection duration affects long-term pricing; a patent expiry could lead to generic competition.
- Formulation innovation (e.g., allergen-free, preservative-free) can justify higher prices.
Regulatory and Healthcare Policy Impact
- Insurance reimbursement policies will influence patient out-of-pocket costs.
- FDA and EMA approval can impact market access and pricing strategies.
- Price controls in certain markets (e.g., Europe) may limit maximum allowable prices.
Key Takeaways
- The market for long-acting nasal allergy medications is growing, with an annual increase driven by rising allergy prevalence.
- "24H Nasal Allergy" is likely positioned as a premium, convenient alternative, influencing pricing structures.
- Entry timing, patent longevity, and competitive dynamics will shape its pricing trajectory.
- Estimated monthly treatment costs range from $20 in early stages to around $32 by 2030.
FAQs
1. What factors influence the pricing of allergy medications?
Pricing is affected by formulation innovation, patent status, regulatory approval, competitive landscape, reimbursement policies, and manufacturing costs.
2. How do long-acting nasal allergy drugs compare prices-wise to short-acting variants?
Long-acting formulations generally carry a 20–30% premium over short-acting versions due to convenience and efficacy claims.
3. What is the regulatory pathway for a novel nasal allergy medication?
Typically involves FDA 505(b)(2) for new formulations of existing drugs or full NDA for novel compounds, plus EMA marketing authorization.
4. How does market penetration vary across regions?
Developed markets like North America and Europe exhibit higher adoption rates, with prices influenced by insurance coverage; emerging markets show rapid growth potential with price sensitivity.
5. When could patent expiry impact the market for 24H Nasal Allergy?
Patents typically last 10–12 years from filing. Expiry could lead to generic competition, decreasing prices by 50% or more within 2–3 years afterward.
Sources:
[1] Grand View Research, "Allergic Rhinitis Market Size & Share," 2022.
More… ↓
