Key Takeaways

TCO Has Permanently Replaced PPV as the Operating Metric. Purchase Price Variance (PPV) models are inadequate for pharmaceutical procurement. Inventory carrying costs in pharma average 19.6% of inventory value annually, and cold chain logistics consume capital at a rate that makes the visible acquisition cost less than 60% of the total lifecycle expense. Any procurement team still reporting primarily on PPV is measuring the wrong variable.
DSCSA and BIOSECURE Have Become Market-Access Prerequisites, Not Compliance Checkboxes. The Electronic Product Code Information Services (EPCIS) standard and the legal exclusion of designated ‘biotechnology companies of concern’ now determine who can participate in the U.S. drug supply chain as an authorized trading partner. Regulatory adherence has become a primary qualification criterion, ahead of price.
Patent Intelligence Determines Sourcing Timelines, Not the Reverse. In the generics market, procurement decisions on API sourcing are binary if they are not informed by real-time patent expiration data, Paragraph IV certification windows, and ‘skinny label’ use-code maps. Platforms like DrugPatentWatch exist precisely to close this gap. Filing a substantially complete ANDA with a Paragraph IV certification without a secured, non-infringing API supply is a company-ending mistake.
Single-Source Efficiency Is Finished as a Strategy. The Pfizer Rocky Mount tornado (July 2023) and the Change Healthcare ransomware attack (February 2024) are not outliers. They are the new operating environment. Procurement leaders are building geographic and vendor redundancy into their sourcing models, using digital twins for scenario simulation, and formally documenting concentration risk in geopolitical hubs.
Procurement Owns Corporate Net Zero. Scope 3 emissions — indirect emissions from purchased goods and services — account for 90-92% of a pharmaceutical company’s total carbon footprint. No R&D or manufacturing efficiency program will get a company to net zero without procurement-binding supplier codes of conduct, primary emissions data collection, and green chemistry sourcing mandates.
1. The New Economic Architecture: From Price to Total Cost of Ownership
1.1 Why Purchase Price Variance (PPV) Is the Wrong Metric
The institutional history of pharmaceutical procurement is rooted in a simple transactional logic: source the Active Pharmaceutical Ingredient (API) or excipient at the lowest unit cost and pass the savings through to margins. That logic held when supply chains were short, regulatory requirements were manageable, and biologics were a rounding error in the product mix. None of those conditions apply today.
Purchase Price Variance measures the difference between the actual price paid and a standard benchmark price. It rewards buyers who negotiate favorable unit costs, which creates a perverse incentive to select suppliers who offer the lowest invoice price regardless of their regulatory track record, geographic risk profile, or logistical reliability. A supplier who delivers API at 15% below the market average but requires three batch retests per year, ships from a region with geopolitical exposure, and cannot produce valid EPCIS data on shipment day is not cheaper. It is more expensive by a significant margin once the full cost architecture is modeled.
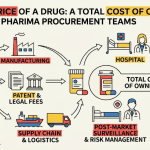
Total Cost of Ownership (TCO) replaces PPV as the operating metric by capturing every cost that a procured item generates across its lifecycle: qualification, acquisition, logistics, quality assurance, inventory holding, regulatory compliance, and disposal. The transition from PPV to TCO is not a philosophical shift; it is a practical necessity in an industry where the hidden costs of procurement routinely exceed the invoice price.
1.2 Deconstructing Pharmaceutical TCO: A Full Component Map
Pre-Acquisition Costs
Before a single purchase order is issued, pharmaceutical procurement incurs substantial costs. Qualifying a new API supplier is a multi-stage exercise involving desk-based audits, site qualification visits (typically billed at $2,000-$5,000/day for a qualified auditor plus travel), stability batch testing across ICH conditions (six months minimum for primary stability data), and potential regulatory filings. If the supplier holds a Drug Master File (DMF) that requires a Type II amendment to reflect the new customer’s process, that filing incurs FDA review time of 12-24 months. The pre-acquisition cost for a critical API supplier qualification routinely exceeds $150,000 before a single kilogram is purchased.
These costs are real and must be amortized over the expected lifetime of the sourcing relationship. This is why switching suppliers mid-contract, even when a competitor offers a lower unit price, is rarely economically rational when a full TCO model is applied.
Acquisition Costs and Landed Cost Calculations
The invoice price is only the starting point. International API transactions require a full ‘landed cost’ calculation that incorporates ocean or air freight (API shipments via air freight typically cost 8-12x sea freight per kilogram), insurance, customs duties under the applicable Harmonized Tariff Schedule (HTS) code, customs brokerage fees, and currency hedging costs if the transaction is denominated in a foreign currency. Under Section 301 tariffs on Chinese goods, API imports from China face additional duties of 7.5-25% depending on the chemical classification. These tariffs have materially eroded the cost advantage of Chinese API manufacturers and are a live variable in any landed cost model.
Operating Costs: The Undercount Problem
Operating costs are the component most frequently underweighted in pharmaceutical TCO models. For a raw material, these include the labor time for receiving inspection, sampling, QC testing (HPLC, NIR, dissolution), documentation review, and the warehouse handling to move material from quarantine to released inventory. For a reagent used in a biological manufacturing process, a failed in-process test that triggers a deviation investigation can cost $50,000-$200,000 in direct investigation labor, batch hold costs, and potential batch loss before the root cause is traced to a sub-specification material.
For capital equipment procurement, energy consumption deserves particular scrutiny. Freeze dryers (lyophilizers) used in biologic manufacturing are among the most energy-intensive assets in a pharmaceutical facility, consuming 10-15 kWh per kilogram of product dried. Over a 15-year asset life, energy costs routinely exceed the original capital purchase price by a factor of two.
Quality and Risk Costs: The Largest and Least Predictable Variable
This is where TCO models diverge most sharply from PPV-based thinking. The FDA’s Enforcement Action database is a publicly searchable record of Warning Letters, Import Alerts, and consent decrees. A supplier operating under a consent decree cannot export product to the U.S. A supplier subject to an Import Alert 66-40 (failure to comply with GMP) has product detained at the port of entry until the alert is lifted — a process that takes 12-36 months on average.
Procurement teams must quantify this risk. A risk premium framework assigns each supplier a probability-weighted cost of supply disruption based on factors including its FDA inspection history (count of 483 observations in the past three years), the FDA’s Data Integrity indicator (presence or absence of data integrity findings, which typically accompany the most severe enforcement actions), country-of-manufacture risk (India and China account for the majority of Warning Letters issued to API manufacturers), and concentration of manufacturing within a single site.
The financial cost of a Class I recall — the most severe category, covering products that could cause serious adverse health consequences — averages $150 million when direct recall costs, legal liability, lost sales, and brand damage are included. Even a Class III recall (unlikely to cause adverse health consequences) generates $5-10 million in direct costs. TCO models that ignore recall probability are not TCO models; they are invoice trackers with better formatting.
End-of-Life Costs
Pharmaceutical waste disposal is regulated under the Resource Conservation and Recovery Act (RCRA) and, for controlled substances, under DEA regulations. The destruction of a batch of a Schedule II controlled substance API requires DEA Form 41, witnessed destruction, and chain-of-custody documentation. Third-party destruction contractors charge $5-$20 per kilogram for hazardous pharmaceutical waste, with surcharges for highly potent or cytotoxic compounds. A procurement team holding 500kg of expired API from a bulk purchase made for volume discount purposes is paying those destruction costs in addition to the carrying costs accrued over the expiration period.
1.3 Inventory Carrying Costs: The 19.6% Drain
Pharmaceutical inventory carrying costs average 19.6% of inventory value annually. For a company carrying $100 million in inventory, that is $19.6 million per year in direct cost — before a single unit expires or is recalled.
The four components break down as follows. Capital cost, the opportunity cost of the cash invested in inventory rather than deployed in R&D or returned to shareholders, runs 6-12% annually depending on the company’s weighted average cost of capital (WACC). Service costs — insurance and taxes on inventory assets — add 3-9%, elevated in pharma by the high liability profile of the products. Storage costs for standard ambient warehouse space run 2-5%, but cold chain requirements inflate this materially; refrigerated warehouse space (2-8°C) typically costs three to four times ambient space per square foot, and cryogenic storage (-70°C to -80°C) for biologics and cell therapies adds another order of magnitude. Risk costs — obsolescence, expiration, pilferage, and damage — add 6-12%, with the ‘patent cliff’ adding a unique dimension: a generic launch can instantly render finished goods inventory economically obsolete even before it expires.
The procurement implication is direct. A volume purchase of API at a 12% discount that results in 12 months of inventory coverage is economically neutral at best and loss-generating at worst when the 19.6% annual carrying cost is applied. A lean, 90-day inventory model at full price is frequently cheaper on a TCO basis. This calculation must be performed on each API line rather than applied as a blanket policy, because some APIs have long lead times or constrained supply that justify strategic buffer stock.
1.4 Cold Chain Economics and the Biologic Procurement Premium
The commercial shift toward biologics, cell and gene therapies (CGTs), and mRNA-based products has created a procurement sub-discipline around cold chain management. These products demand temperature ranges of 2-8°C (standard refrigerated), -20°C (frozen), or -70°C to -196°C (cryogenic), with zero tolerance for temperature excursions.
The capital expenditure (CapEx) burden is significant. An ultra-low temperature (ULT) freezer capable of holding 700 liters at -80°C costs $8,000-$15,000. A cryogenic vapor-phase liquid nitrogen storage system for cell therapy products costs $25,000-$60,000 per unit, with ongoing liquid nitrogen consumption running approximately $2,000-$4,000 per year per unit. A dedicated cold chain packaging evaluation for a new biologic route typically takes 6-9 months of seasonal testing (hot summer and cold winter profiles) before packaging can be validated.
On the logistics side, the 3PL selection decision for cold chain products deserves TCO modeling that most procurement organizations have not built. The key variables are lane-specific temperature excursion rates (which responsible 3PLs disclose), the cost of a temperature excursion (which, for a biologic drug product, can be $50,000-$2 million per shipment), the cost of monitoring infrastructure (continuous temperature loggers run $50-$150 per shipment for single-use devices, or can be bundled into 3PL contracts for multi-use IoT loggers), and the 3PL’s ability to provide real-time temperature data via API feed for digital twin integration.
Investment Strategy Note: Cold chain logistics infrastructure is a capital-intensive sector with durable demand driven by biologic growth. Companies with material exposure to cell and gene therapy logistics — including specialized 3PLs and packaging firms — carry a structural tailwind as biologic pipelines mature. Procurement intelligence on which CDMOs are winning biologic manufacturing contracts is a leading indicator of demand for their logistics partners.
1.5 Key Takeaways: Section 1
TCO is not an accounting exercise; it is a risk-adjusted sourcing framework. The 19.6% annual carrying cost benchmark is a hard financial fact that should govern volume decisions. Supplier qualification costs must be amortized into the sourcing relationship economics before switching analysis is conducted. Cold chain logistics costs for biologics and CGTs require dedicated modeling separate from standard ambient supply chain economics.
2. Regulatory Compliance as a Procurement Lever
2.1 The Drug Supply Chain Security Act (DSCSA): Full Implementation Architecture
The DSCSA, enacted in 2013 under Title II of the Drug Quality and Security Act, mandated a decade-long phased implementation of electronic, interoperable track-and-trace for all prescription drug packages in the U.S. supply chain. As of November 2024, the final phase is in effect: all trading partners must transact in EPCIS-compliant serialized data or they lose their Authorized Trading Partner (ATP) status.
The ATP Legal Framework
‘Authorized Trading Partner’ is a legally defined status under 21 U.S.C. § 360eee. It requires that manufacturers, wholesale distributors, dispensers, and third-party logistics providers hold a valid license under applicable state and federal law and are registered with the FDA. Procurement contracts must verify ATP status annually. Sourcing from a non-ATP entity is not a paperwork deficiency; it is a federal violation carrying civil monetary penalties up to $500,000 and potential license revocation. Supply agreements must include ATP status as a condition of contract, with self-certification requirements and right-to-verify clauses.
EPCIS 1.2 and the Serialization Stack
EPCIS (Electronic Product Code Information Services) version 1.2 is the GS1 standard that DSCSA mandates for data exchange. Every prescription drug package must carry a National Drug Code (NDC), serial number, lot number, and expiration date encoded in a 2D barcode (GS1 DataMatrix). When a package changes hands, the seller must generate Transaction Information (TI), a Transaction History (TH), and a Transaction Statement (TS) — collectively the ‘3Ts’ — in EPCIS format.
The ‘Data, No Product’ problem is the most operationally disruptive failure mode. When a truckload of product arrives at a wholesale distribution center without accompanying EPCIS data, the receiving system flags every item as ‘suspect product.’ The product cannot be put into sellable inventory until the data discrepancy is resolved. It sits in physical quarantine accruing storage costs, and if the shelf life is short, it is at risk of reaching its expiration date before the data issue is resolved. This is not a theoretical problem; in the first months following full DSCSA implementation, industry groups reported exception rates of 3-8% on inbound shipments from smaller manufacturers who had not fully upgraded their serialization systems.
Procurement contracts must therefore include explicit penalty clauses for serialization failures. A well-drafted clause defines a resolution SLA (typically 24-48 hours for data exceptions), specifies that the supplier bears all costs associated with quarantine, demurrage, and redelivery, and grants the buyer the right to return product and cancel the purchase order if resolution is not achieved within the SLA window.
Exception Handling and the ‘Illegitimate Product’ Trigger
The DSCSA requires trading partners to investigate ‘suspect product’ (product reasonably suspected of being counterfeit, diverted, stolen, or intentionally adulterated) and ‘illegitimate product’ (suspect product that has been confirmed to be one of these things). An illegitimate product finding triggers a mandatory notification to the FDA within 24 hours. Procurement must establish formal exception handling protocols with each trading partner, defining the investigative workflow, communication responsibilities, and decision tree for FDA notification. These protocols belong in the Quality Agreement, not in the Supply Agreement.
2.2 The BIOSECURE Act: Geopolitical Decoupling in Operational Detail
The BIOSECURE Act was enacted in January 2026 after a prolonged legislative process. It prohibits federal agencies from contracting with ‘biotechnology companies of concern’ and restricts federal grantees — including NIH-funded academic centers and biopharma companies receiving federal grants — from using the equipment or services of designated entities.
The Designated Entity Problem
The entities targeted by BIOSECURE legislation include major Chinese CDMOs and genomic data companies. WuXi AppTec and its affiliates represent the sharpest operational problem for the global biopharma industry. WuXi AppTec operates as a contract manufacturing organization (CDMO) for small molecules and as a contract research organization (CRO) for drug discovery services. Its affiliate WuXi Biologics handles biological drug substance manufacturing. Estimates suggest WuXi entities have manufacturing relationships with a significant fraction of the top 20 global pharmaceutical companies and hundreds of smaller biotechs. BGI Genomics, MGI Tech, and Complete Genomics are designated primarily for their genomic data activities.
The ‘equipment or services’ language is broad. A university genomics core facility using BGI sequencing equipment could jeopardize its NIH grant eligibility. A biotech company using WuXi’s chemistry services for early-stage lead optimization could be ineligible for federal contracts even if it never uses WuXi for clinical manufacturing. Procurement and legal must conduct a deep-tier audit that covers not just direct vendor relationships but the equipment installed in third-party CRO and CDMO facilities where the company’s work is performed.
The Technology Transfer Problem
The grandfathering clause in the enacted version of BIOSECURE gives existing contracts until 2032 to wind down. This sounds generous until the technology transfer timeline is mapped out. Moving a biologic drug substance process from one CDMO to another requires process characterization at the new site, engineering runs, GMP manufacturing of comparability batches, analytical comparability studies across a comprehensive attribute panel (potency, purity, glycosylation profile, higher-order structure), and a Prior Approval Supplement (PAS) or CBE-30 submission to the FDA. The full timeline for a complex biologic technology transfer is 36-60 months. Companies that begin their BIOSECURE transition planning in 2026 are running a 12-24 month margin relative to the deadline; companies waiting until 2028 or later will face a compressed and expensive transition.
The CDMO Capacity Crunch
The non-Chinese CDMO industry does not have unlimited capacity to absorb the volume of work currently performed by WuXi and other designated entities. European CDMOs (Lonza, Rentschler, Samsung Biologics’ European operations, Novasep) and U.S.-based manufacturers (Emergent BioSolutions, Catalent, Grand River Aseptic Manufacturing) are already operating at high utilization rates. Demand from BIOSECURE-driven reshoring will tighten capacity further and allow CDMOs to negotiate less favorable terms for new clients. Procurement teams entering negotiations for CDMO slots in 2026-2028 should expect premium pricing and extended lead times relative to the 2020-2023 market.
Investment Strategy Note: BIOSECURE is a structural demand driver for non-Chinese CDMOs. Lonza (LONN.SW), Samsung Biologics (207940.KS), and Wuxi Biologics’ competitors in Europe and the U.S. are direct beneficiaries. The capacity constraint implies pricing power for established Western CDMOs over the 2026-2030 period. Investors tracking CDMO contract announcements — visible through regulatory filings, manufacturing site authorizations, and Drug Master File submissions — can identify which players are capturing the reshoring volume first.
2.3 The Bifurcation of Agreements: Quality Agreement Architecture
The separation of Quality Agreements from Supply Agreements is mandated by the FDA’s 2016 Quality Agreements Guidance for Industry and equivalent EMA guidance. The principle is that quality decisions — batch release, deviation closure, raw material rejection — must be made on scientific and regulatory grounds without contamination by commercial considerations.
Quality Agreement Core Elements
A well-constructed Quality Agreement for a CDMO or API supplier relationship must define ownership of every GMP-relevant activity using a RACI (Responsible, Accountable, Consulted, Informed) matrix. The key activities include raw material release, in-process testing, batch manufacturing record review, batch disposition and release, deviation investigation and CAPA closure, annual product review compilation, and change control management.
Change control is the highest-stakes element for API sourcing. A supplier who changes their starting material source, introduces a new synthesis step, switches an excipient, or modifies an analytical testing method without prior notification and approval from the buyer has compromised the buyer’s regulatory filings. The buyer’s Drug Application (NDA, ANDA, BLA) is filed with specific process and quality information; unauthorized changes at the supplier constitute a potential regulatory violation that can halt product marketing.
The Quality Agreement must specify the change notification requirements in granular detail: what types of changes require prior approval versus pre-implementation notification, what data the supplier must provide to support the change, and what the buyer’s review timeline is. The FDA’s 21 CFR 314.70 and 314.97 define the change categories for drug products; an equivalent framework must be negotiated into the Quality Agreement for raw material suppliers.
The ‘No Commercial Terms’ Principle
Best practice — and increasingly a regulatory expectation — is that Quality Agreements contain zero commercial terms. No pricing, no liability caps, no indemnification language, no payment terms. This separation protects the integrity of quality decisions. A quality manager who finds a batch defective but knows the Supply Agreement imposes a $500,000 penalty for batch rejection is in a conflict of interest. The Quality Agreement must be drafted so that quality decisions stand independently of their financial consequences.
2.4 Key Takeaways: Section 2
DSCSA full implementation has made serialized data exchange a transactional prerequisite; procurement contracts that do not specify EPCIS compliance obligations and exception-handling SLAs are incomplete. The BIOSECURE Act requires immediate deep-tier supply chain audits and technology transfer planning with a 36-60 month horizon. Quality Agreements must be isolated from commercial terms and must include granular change control notification requirements to protect regulatory filing integrity.
3. Intellectual Property Valuation and Competitive Sourcing
3.1 The IP Asset Map: What Procurement Must Understand
Intellectual property is not just a legal matter for pharma companies — it is the primary determinant of market entry timing, revenue duration, and sourcing strategy for both innovators and generic manufacturers. A procurement team that does not understand the IP landscape around the drugs it sources is flying blind on the most consequential variable in its planning horizon.
For innovator companies, the IP asset portfolio around a drug product typically includes a composition of matter (CoM) patent covering the API structure, one or more formulation patents covering drug delivery systems, method-of-use patents covering specific therapeutic indications, process patents covering the synthesis route, and potentially a polymorph patent covering a specific crystalline form. Each of these patents appears in the FDA’s Orange Book if it meets the listing criteria, and each represents a separate layer of protection that generic entrants must address.
Composition of Matter Patents: The Crown Jewel
The CoM patent is the most durable and defensible form of pharmaceutical IP. It protects the molecule itself, regardless of indication, formulation, or dose. For a blockbuster drug, the CoM patent is typically filed during early clinical development (Phase I or before), giving it a 20-year statutory term from filing. After accounting for Patent Term Extension (PTE) under 35 U.S.C. § 156, which can add up to 5 years for the time spent in regulatory review, and any Pediatric Exclusivity extension adding 6 months under the Best Pharmaceuticals for Children Act, the effective market exclusivity period for a blockbuster can extend to 27-28 years from the initial patent filing.
IP Valuation for Procurement Strategy
From the generic procurement perspective, the CoM patent expiration date (adjusted for PTE and exclusivity extensions) defines the earliest possible ANDA approval date for a product without Paragraph IV challenge. Procurement must map this date precisely because the API sourcing timeline runs backward from it. To file a substantially complete ANDA, the generic company needs a validated API synthesis route, confirmed DMF support from the API supplier, and at minimum 3 months of accelerated stability data (with 6 months preferred). Working backward, procurement must have a qualified, non-infringing API supplier locked in at least 18-24 months before the target ANDA filing date.
DrugPatentWatch’s Orange Book data provides this patent mapping in real time, including the specific Use Codes attached to each listed patent. This granular data is essential for identifying whether a skinny label carve-out is feasible or whether a Paragraph IV certification is the only viable path to market.
3.2 Paragraph IV Strategy: The Full Operational Playbook
The Hatch-Waxman Act’s Paragraph IV mechanism is the central legal instrument of generic market competition in the U.S. A Paragraph IV certification asserts that a listed patent is invalid, unenforceable, or will not be infringed by the proposed generic product. Filing it is an act of patent infringement that triggers a 30-month stay on ANDA approval (unless the court rules in favor of the generic company sooner).
The 180-Day Exclusivity Prize
The first generic company to file a substantially complete ANDA containing a Paragraph IV certification for a given drug-patent combination receives 180 days of market exclusivity. During this period, the FDA will not approve any other generic ANDA for that drug. Given that generic drugs typically capture 80-90% of brand unit volume within 90 days of launch, and that the first-to-market generic often prices at 50-80% of the brand price before a second generic enters, the 180-day exclusivity window can generate $100 million to over $1 billion in revenue depending on the brand’s market size.
This is not an abstraction for procurement teams. To win the first-to-file race, the generic company must have an ANDA ready to submit before competitors. Every day of delay in the API sourcing process is a potential day of exclusivity lost. Procurement’s ability to identify, qualify, and lock in an API supplier ahead of the patent expiration curve is a direct financial performance driver.
What ‘First-to-File’ Demands of Procurement
Winning the first-to-file race requires the API supplier to meet several criteria simultaneously. The supplier must be technically capable of producing the API to required purity specifications — which for complex molecules with chiral centers or polymorph sensitivity can be demanding. The supplier must hold or be willing to file a Drug Master File (DMF) with the FDA; a Type II DMF for an API covers the manufacturing process, specifications, and analytical methods and must be current and not subject to outstanding FDA requests. The supplier’s synthesis route must be non-infringing relative to the innovator’s process patents, which requires a freedom-to-operate (FTO) analysis conducted by the generic company’s IP counsel before API sourcing is finalized. The supplier must be audit-ready and must have a satisfactory FDA inspection history, because an API manufactured at a facility under Import Alert or Warning Letter cannot support an approved ANDA.
Monitoring the Paragraph IV Pipeline
For institutional investors and procurement planners alike, the Paragraph IV pipeline is a real-time indicator of competitive pressure on brand franchises. The FDA publishes Paragraph IV certification notifications in the Federal Register. Each notice discloses the ANDA filer, the referenced NDA, the specific patents being challenged, and whether any other ANDA filers have shared ‘first-to-file’ status (which splits the 180-day exclusivity between co-filers). Tracking this data identifies which brands are under imminent competitive attack and which APIs will be in sudden high demand among generic procurement teams.
Investment Strategy Note: A brand drug receiving its first Paragraph IV certification is a defined-duration revenue event for the innovator. The 30-month stay provides a window, but the subsequent generic launch typically generates rapid erosion. Short positions on innovator companies with major Paragraph IV challenges and long positions on the generic filer with 180-day exclusivity are a well-established trade in pharma equities. The risk variable is litigation outcome: if the court upholds the brand patents within the 30-month stay, the generic launch is blocked and the innovator retains pricing power. DrugPatentWatch’s litigation tracking data is the primary source for monitoring case progression.
3.3 Evergreening: The Innovator’s Procurement Defense Playbook
Evergreening is the collection of strategies by which innovator companies extend market exclusivity beyond the expiration of the original composition of matter patent. From the procurement perspective, evergreening extends the period during which sourcing must accommodate brand-price tiers and delays the window for generic API procurement.
The Technology Roadmap for Evergreening
Formulation Innovation: Transitioning a drug from an immediate-release tablet to an extended-release formulation generates a new patent on the delivery technology and allows the innovator to file new Orange Book patents. The brand is then marketed as the extended-release version (often with a new trade name), and the innovator discontinues the original formulation. Generic companies that have been developing an IR formulation face the choice of pursuing the now-discontinued original market or developing an ER equivalent, which requires new formulation development and stability testing. AbbVie’s transition from Humira (adalimumab) subcutaneous injection to Skyrizi and Rinvoq was partly a portfolio evolution strategy that shifted the center of gravity of the franchise to newer, more heavily patented assets before Humira’s biosimilar competition intensified.
Polymorph and Salt Form Patents: The FDA approves a drug product based on the specific polymorphic form and salt form described in the application. A patent on a specific polymorph of a drug can block generic competition if the generic cannot demonstrate bioequivalence using a different polymorph, or if the patented polymorph is necessary for the approved formulation performance. Generic procurement teams must assess polymorph patent exposure before committing to an API synthesis route.
Pediatric Exclusivity as an Evergreening Tool: The Best Pharmaceuticals for Children Act grants 6 months of additional exclusivity to any drug for which the innovator conducts a pediatric study requested by the FDA. This extension applies to all Orange Book patents, not just the patent for the studied indication. For a blockbuster with multiple patents, pediatric exclusivity can delay generic entry by 6 months across the entire patent portfolio, representing hundreds of millions of dollars in protected revenue.
Method-of-Use Patents and New Indication Filing: Filing a new NDA supplemental indication for a drug generates new method-of-use patents and potentially new regulatory exclusivity periods (3 years for a new clinical investigation supplement). Generic companies using skinny labels to avoid the new indication must carefully track what use codes are attached to which Orange Book patents and whether FDA has updated the use code list following the new supplemental approval.
3.4 Skinny Labeling: The Legal Minefield in Detail
Section viii of the Hatch-Waxman Act allows a generic manufacturer to seek approval for less than all the patented uses of a reference listed drug by carving out the patent-protected indications from the generic label. This mechanism is supposed to allow competitive market entry for unpatented indications while respecting method-of-use patent protection.
The Induced Infringement Problem
The legal theory of induced infringement holds that a party who actively encourages another party to infringe a patent can be liable for that infringement even if the inducing party does not itself directly infringe. In the skinny label context, the courts have applied this theory to find generic manufacturers liable when their product labeling, press releases, investor materials, or marketing communications direct patients or physicians toward the patented indication, even if the patented indication has been carved out of the FDA-approved label.
The GSK v. Teva litigation (involving carvedilol and its patented heart failure indication) established that a generic’s carve-out label did not insulate it from induced infringement liability because Teva’s marketing materials referenced the heart failure use. The case produced contradictory jury verdicts across multiple trials and a tortuous appeals process, culminating in a damages award that was remanded and retried multiple times. The Amarin v. Hikma litigation (involving icosapentaenoic acid / Vascepa) added another dimension: Hikma’s generic label carved out Amarin’s patented cardiovascular outcomes indication, but Amarin argued that Hikma’s press releases highlighting the total market for the drug (which was dominated by the patented indication) constituted active inducement.
Procurement Implications: Contract Architecture for Skinny Label Products
Any procurement of a product bearing a skinny label — whether by a distributor, health system, pharmacy benefit manager (PBM), or specialty pharmacy — carries a non-trivial induced infringement exposure. The procurement contract must include several protective elements.
Indemnification language must require the generic manufacturer (or authorized distributor) to defend and indemnify the buyer against any patent infringement claim arising from the use of the product. The scope of this indemnification should be defined to cover claims arising from the buyer’s ordinary course distribution activities, with carve-outs only for the buyer’s own affirmative marketing of the product for the patented indication.
Promotional restriction clauses must prohibit the seller from providing the buyer with marketing materials, formulary recommendations, or clinical support materials that reference the patented indication. The buyer’s own formulary placement decisions and PBM management activities must be reviewed by IP counsel before any carvedilol-equivalent product is added to a formulary with substitution provisions.
The Supreme Court’s current posture on induced infringement in the skinny label context is evolving. Procurement legal teams must track the cert petition activity in cases arising from the circuit courts, as a Supreme Court decision clarifying the standard for induced infringement in the Hatch-Waxman context would have immediate implications for every skinny label product in commercial distribution.
3.5 Key Takeaways: Section 3
Patent intelligence is a sourcing discipline, not just a legal function. The 180-day Paragraph IV exclusivity window is a quantifiable revenue target that procurement must support by securing qualified API supply at least 18-24 months before the target ANDA filing. Evergreening strategies — extended-release reformulation, polymorph patents, pediatric exclusivity, new indications — extend the competitive timeline on each brand franchise and must be modeled into generic sourcing forecasts. Skinny label procurement requires indemnification clauses and promotional restriction covenants to manage induced infringement exposure.
4. The Biosimilar Procurement Frontier
4.1 Biosimilar Interchangeability: The Regulatory Standard and Its Procurement Consequences
The BPCIA’s biosimilar framework created a tiered approval system. An approved biosimilar has demonstrated that it is ‘highly similar’ to the reference biologic with ‘no clinically meaningful differences’ in safety, purity, or potency. An interchangeable biosimilar has met an additional standard: it must produce the same clinical result as the reference product in any given patient, and if the product is administered more than once, the risk of alternating between the interchangeable and the reference must be no greater than using the reference alone without alternation. Interchangeability allows a pharmacist to substitute the biosimilar for the reference product without physician intervention, which is the critical enabling condition for formulary-level substitution.
As of early 2026, the FDA has designated fewer than 15 biosimilars as interchangeable. The insulin market has seen the most aggressive interchangeability designations, with products referencing Lantus (insulin glargine) and Humalog (insulin lispro) receiving interchangeable status. Humira (adalimumab) biosimilars have achieved interchangeable status from several manufacturers, producing the most complex biosimilar market in U.S. history — with over a dozen competitors offering both citrate-free and high-concentration formulations.
Procurement for health systems, PBMs, and specialty distributors must distinguish between biosimilars and interchangeable biosimilars in formulary design. Non-interchangeable biosimilars cannot be automatically substituted at the pharmacy level in states that have not passed specific substitution permissibility statutes. Procurement contracts for biosimilar products should specify whether the product is being purchased as an interchangeable and should include provisions for updating the contract if the interchangeability designation is granted or revoked.
4.2 IP Valuation in the Biosimilar Context: The Reference Biologic’s Patent Thicket
Reference biologic products are typically protected by a patent thicket — a dense cluster of overlapping patents covering the molecule, the manufacturing process, the formulation, the delivery device, and the methods of use. AbbVie’s Humira (adalimumab) IP portfolio has been cited as the most extensive pharmaceutical patent thicket in history, with over 130 patents listed in the Purple Book (the biologic equivalent of the Orange Book) at various points before biosimilar entry. The manufacturing process patents alone — covering cell culture conditions, purification steps, and formulation parameters — were sufficient to delay biosimilar market entry in the U.S. until 2023, despite Humira biosimilars being available in Europe from 2018.
For biosimilar manufacturers, the procurement challenge mirrors the small molecule Paragraph IV situation but with greater complexity. The BPCIA’s ‘patent dance’ requires the biosimilar applicant to disclose its manufacturing process to the reference product sponsor in a confidential exchange, after which the sponsor identifies the patents it believes are infringed. The litigation that follows covers not just the reference molecule’s composition patents but also process and formulation patents that the biosimilar manufacturer must design around. Procurement of raw materials for biosimilar manufacturing must therefore account for the possibility that a manufacturing process change required to design around a process patent will necessitate a supplemental BLA submission and a new comparability exercise.
Investment Strategy Note: The Humira biosimilar market is a case study in how patent thicket deconstruction affects pricing. The entry of multiple citrate-free, high-concentration biosimilars priced at 5-85% discounts to Humira’s list price created an immediate formulary management opportunity for PBMs and health systems. Procurement teams that moved early to establish biosimilar preferred tiers generated measurable formulary savings within 12-18 months of biosimilar entry. The pattern will repeat with Keytruda (pembrolizumab), Opdivo (nivolumab), and Eylea (aflibercept) as their patent thickets thin in the late 2020s.
4.3 Key Takeaways: Section 4
Biosimilar interchangeability is a binary regulatory designation that determines whether pharmacist-level substitution is permissible — this must be specified in procurement contracts. The reference biologic patent thicket defines the timeline and litigation risk for biosimilar market entry. The Humira biosimilar market is the current benchmark for post-thicket price dynamics, and the same pattern will apply to monoclonal antibody blockbusters approaching patent expiration in the late 2020s.
5. Supply Chain Resilience and Risk Mitigation
5.1 Concentration Risk: The Rocky Mount Anatomy of a Single-Point Failure
On July 19, 2023, an EF3 tornado struck Pfizer’s manufacturing complex in Rocky Mount, North Carolina. The facility’s production lines, housed in hardened structures, survived largely intact. The adjacent warehouse, storing approximately $814 million in finished goods and raw materials, was destroyed. The site accounted for roughly 25% of all sterile injectable drugs used in U.S. hospitals — including critical-care staples like lidocaine, epinephrine, morphine, and neuromuscular blocking agents.
The immediate consequence was that Pfizer placed dozens of products on ‘strict allocation,’ rationing available inventory to prevent hoarding while the facility resumed operations. Hospital pharmacy directors were forced to implement substitution protocols, cancel elective surgical procedures that required unavailable agents, and draw down strategic reserves. The FDA issued emergency guidance allowing importation of foreign-manufactured alternatives and expedited manufacturing supplements for other producers.
The Rocky Mount event did not reflect negligence. Pfizer’s operations at the site met every applicable GMP standard. The failure was structural: the concentration of so much critical drug production in a single geographic location created a vulnerability that no quality system could address. The lesson for procurement is that concentration risk is a portfolio-level problem requiring a portfolio-level solution. The U.S. Pharmacopeia’s Medicine Supply Map tool assigns vulnerability scores to drugs based on the number of manufacturers, geographic diversity of those manufacturers, and manufacturing complexity. Procurement teams should run their critical formulary items through this tool and treat high-vulnerability scores as sourcing deficiencies requiring active remediation.
5.2 Cybersecurity: Change Healthcare and the Vendor-as-Entry-Vector Problem
In February 2024, the BlackCat/ALPHV ransomware group exploited a compromised credential set to access Change Healthcare’s network via a Citrix remote access portal that lacked multi-factor authentication. Change Healthcare, a subsidiary of UnitedHealth Group, processes roughly 15 billion healthcare transactions annually and handles claims for approximately one-third of all U.S. patients. The attack took Change Healthcare’s systems offline for weeks, halting pharmacy claims adjudication, prior authorization processing, and revenue cycle operations across thousands of healthcare facilities.
The financial impact was extraordinary. UnitedHealth Group disclosed over $5.4 billion in total estimated losses and payments related to the attack across 2024 and into 2025. Downstream pharmaceutical distributors and specialty pharmacies lost cash flow from claims processing delays; some independent pharmacies were forced to extend credit to patients or absorb drug costs pending claims resolution.
The procurement risk lesson is that vendor cybersecurity posture is a procurement responsibility, not an IT afterthought. Any vendor with access to systems that touch prescription drug ordering, claims processing, or logistics coordination is an entry vector into the pharmaceutical supply chain. Procurement must require cybersecurity assessments of key vendors using standardized frameworks (NIST CSF, SOC 2 Type II audits, or the HECVAT questionnaire for healthcare-specific environments). The specific controls most predictive of a successful ransomware defense are multi-factor authentication on all remote access, privileged access management (PAM) to limit lateral movement inside the network, and tested, offline, air-gapped backup systems.
The Cencora Breach: Third-Party Patient Data Risk
In February 2024, Cencora (the rebranded AmerisourceBergen) experienced a data breach that exposed personally identifiable information and protected health information for patients of at least 27 pharmaceutical manufacturers who used Cencora’s patient support services. Those manufacturers — including Abbvie, Bayer, GSK, Novartis, and Pfizer — faced class-action litigation and regulatory scrutiny arising from their vendor’s breach. The lesson: a HIPAA Business Associate Agreement (BAA) is a legal necessity but provides no technical protection. Procurement must require contractual data minimization obligations (vendors should hold only the patient data required to perform the specific service), data retention limits (data destroyed on schedule after service delivery), and breach notification timelines no longer than 72 hours from vendor discovery.
5.3 Digital Twin Technology: From Concept to Operational Deployment
A pharmaceutical supply chain digital twin is a virtual model of the physical supply network that ingests real-time data from multiple sources — ERP systems, logistics platforms, IoT sensor feeds, external weather and geopolitical risk signals — and runs continuously alongside the physical supply chain to provide monitoring, simulation, and optimization capabilities.
The practical applications are operational, not theoretical. Scenario simulation allows a company to run a ‘port closure’ or ‘major API supplier shutdown’ scenario through the digital twin model to identify which finished goods lines would be impacted first, how many days of inventory exist at each distribution node, and what alternative sourcing or re-routing options are available. Companies that ran rocky-mount-equivalent scenarios through digital twins before July 2023 were better positioned to activate contingency sourcing rapidly because they had already mapped the decision tree.
For cold chain specifically, digital twins integrated with IoT temperature logger data can identify route-level excursion patterns — for example, a recurring mild excursion on a specific airline lane during summer months that is within label but trending toward the specification limit — and trigger preventive packaging upgrades before a full excursion occurs.
The technology investment is meaningful but not prohibitive. Tier 1 pharmaceutical companies (Roche, Pfizer, Novartis) have invested $20-50 million in enterprise digital twin platforms. Mid-market companies can access modular digital twin functionality through supply chain SaaS vendors at a fraction of this cost, with implementation timelines of 6-12 months for a basic but functional supply network model.
5.4 Key Takeaways: Section 5
Geographic concentration of critical drug manufacturing is a patient safety risk and a procurement liability. The USP Medicine Supply Map provides a publicly accessible vulnerability scoring tool that should anchor critical-item sourcing strategy. Vendor cybersecurity posture is a procurement responsibility — MFA, PAM, and tested offline backup systems are the non-negotiable baseline. Digital twins are an operational technology for pharmaceutical supply chain management, not a future aspiration, and are accessible to mid-market companies through SaaS deployment models.
6. Digital Transformation of Procurement Operations
6.1 AI-Assisted Spend Analytics and Demand Forecasting
The pharmaceutical procurement function has historically been data-rich and insight-poor. ERP systems generate enormous transaction data volumes, but the analytical tools available to most procurement organizations until recently were limited to backward-looking spend reports and manual category analysis. Machine learning-based spend analytics platforms (Jaggaer, Coupa, GEP Smart) now enable pattern recognition across multi-year spend data that identifies category concentration risks, price anomaly alerts, and demand seasonality with a precision that manual analysis cannot match.
For demand forecasting, AI-based models trained on historical consumption data, patient epidemiology trends, and disease seasonality patterns (particularly for acute care drugs like antivirals and antibiotics) produce significantly more accurate 12-month demand forecasts than traditional moving average models. Improved forecast accuracy directly reduces safety stock requirements — a material working capital benefit given the 19.6% annual carrying cost.
The integration of external data signals into procurement AI systems is the current frontier. Companies are building models that incorporate FDA drug shortage database updates, weather forecast data (to predict cold chain demand patterns), and shipping index data (Freightos Baltic Index) to anticipate freight cost movements and adjust procurement cadence accordingly.
6.2 Blockchain for DSCSA Compliance
Distributed ledger technology has been piloted extensively for DSCSA compliance since the Act’s passage, and several production deployments are operational. The core use case is creating an immutable, shared record of product movement events that any authorized trading partner can verify without reliance on a centralized intermediary. MediLedger, a permissioned blockchain network, has the largest production deployment in pharma, with members including major wholesalers, manufacturers, and specialty distributors.
The DSCSA compliance advantage of blockchain is the resolution of the ‘Data, No Product’ exception. When a shipment’s EPCIS data is recorded on a shared ledger at the point of manufacture, the receiving trading partner can verify the provenance of a product instantly against the ledger record, rather than waiting for data files to be transmitted through EDI channels. Exception rates drop because the data is available ahead of the physical product, not concurrent with or after it.
6.3 Key Takeaways: Section 6
AI-based spend analytics and demand forecasting are production technologies that reduce safety stock requirements and identify procurement risk concentrations. Blockchain deployments for DSCSA compliance are operational at scale through networks like MediLedger and reduce ‘Data, No Product’ exception rates by making provenance data available before the physical product arrives.
7. ESG: Decarbonization Through the Supply Chain
7.1 The Scope 3 Accounting Problem
Pharmaceutical companies report greenhouse gas emissions in three scopes. Scope 1 covers direct emissions from company-owned facilities and vehicles. Scope 2 covers indirect emissions from purchased electricity and heat. Scope 3 covers all other indirect emissions, including those from upstream supply chain activities (purchased goods and services, capital goods, business travel) and downstream activities (use of sold products, end-of-life treatment).
For a typical pharmaceutical manufacturer, Scope 3 emissions account for 90-92% of the total carbon footprint. Category 1 of Scope 3, purchased goods and services, is by far the largest contributor — meaning that the carbon embedded in the APIs, excipients, packaging materials, and contract manufacturing services that procurement buys is the dominant emission source in the corporate inventory. No internal energy efficiency program, no matter how aggressive, will achieve a net zero target if Scope 3 is not addressed.
This places procurement at the center of corporate climate strategy. Procurement is the function that chooses suppliers and writes the contracts that govern supplier behavior. It is also the function with the commercial leverage to require suppliers to report emissions data and commit to decarbonization targets.
7.2 Primary Data Collection: Why Spend-Based Estimates Are Insufficient
The default approach to Scope 3 Category 1 measurement is the spend-based method: apply an emissions factor (kg CO2e per dollar spent) from a database like Ecoinvent or the U.S. EPA supply chain emission factors to the dollars spent in each procurement category. This method is fast and requires no supplier engagement, but it is systematically inaccurate for pharmaceutical procurement because the emission intensity of pharmaceutical-grade APIs varies enormously by synthesis route in ways that spending-level data cannot capture.
An API synthesized via a traditional multi-step batch chemical process may consume 50-200 kg of solvent per kilogram of product and require energy-intensive distillation and crystallization steps. The same molecule synthesized via a biocatalytic or continuous flow chemistry route may consume 80% less solvent and significantly less energy. Both APIs might cost the same per kilogram, but their embodied carbon is radically different. Spend-based emission factors would assign them the same Scope 3 intensity, which is wrong.
The solution is primary data collection: requiring suppliers to report actual energy consumption, fuel use, and process emissions for the specific product they supply. AstraZeneca’s supplier sustainability program, Novo Nordisk’s ‘Suppliers for Zero’ initiative, and Bristol Myers Squibb’s supplier engagement framework all require primary emissions data reporting from strategic suppliers. The data collection is managed through platforms like EcoVadis or direct supplier questionnaires aligned with GHG Protocol standards. Companies that move from spend-based to activity-based to primary emissions data reduce their Scope 3 measurement uncertainty from ±50% to ±10%, enabling more credible target-setting and disclosure.
7.3 Green Chemistry and API Procurement
Green chemistry — the design of chemical products and processes that minimize or eliminate hazardous substance use and generation — has moved from an academic discipline to a procurement requirement in leading pharmaceutical companies. The American Chemical Society’s Green Chemistry Institute Pharmaceutical Roundtable, which includes major pharma companies and API manufacturers, has developed Pharmaceutical Round Table Solvent Selection Guides that score solvents on waste, environmental impact, health, and safety criteria.
Procurement can specify green chemistry requirements in API sourcing contracts by requiring the supplier to use only solvents in the ‘recommended’ or ‘problematic but acceptable’ tiers of the solvent guide, to disclose Process Mass Intensity (PMI) — the ratio of total mass input to the mass of API produced — as a standard metric in the supplier data package, and to have an active continuous improvement program targeting PMI reduction over the contract term.
Enzymatic synthesis and biocatalysis are the most transformative green chemistry routes for complex API molecules. Codexis, the enzyme engineering company, licenses its enzyme technology to major pharmaceutical manufacturers and CDMOs, enabling biocatalytic synthesis steps that replace traditional chemical synthesis steps requiring harsh conditions and hazardous reagents. Procurement teams sourcing APIs where biocatalytic routes are available should evaluate the environmental footprint difference, as it can be substantial and increasingly counts toward supplier scorecarding.
7.4 Social Responsibility: Supply Chain Due Diligence Laws
The German Supply Chain Due Diligence Act (Lieferkettensorgfaltspflichtengesetz, LkSG), in effect since January 2023, requires German companies with more than 1,000 employees to identify, prevent, and remediate human rights and environmental risks in their supply chains, including Tier 1 suppliers and, where there is substantiated knowledge of violations, deeper supply chain tiers. The EU Corporate Sustainability Due Diligence Directive (CSDDD) extends similar requirements across EU member states with a broader scope and harsher penalties.
For pharmaceutical procurement, the highest-risk areas under these frameworks are mining of raw materials used in pharmaceutical packaging (cobalt in battery-powered cold chain equipment, tungsten in surgical tools), agricultural supply chains for herbal APIs, and labor conditions in API manufacturing in regions with documented labor rights concerns. Procurement must conduct human rights due diligence and document it rigorously — the LkSG’s enforcement authority (BAFA) has issued substantial fines for procedural non-compliance even where no actual violations were identified.
The Pharmaceutical Supply Chain Initiative (PSCI) provides a standardized audit framework that allows pharmaceutical companies to share audit reports across member companies, reducing audit burden on suppliers while maintaining a consistent ethical standard. PSCI audits cover labor rights, health and safety, environmental management, business ethics, and management systems. Requiring PSCI audit compliance as a supplier qualification criterion is increasingly standard practice for major pharmaceutical manufacturers.
7.5 Key Takeaways: Section 7
Scope 3 Category 1 emissions dominate the pharmaceutical carbon footprint, and spend-based measurement is too inaccurate to support credible disclosure or target-setting. Primary data collection from suppliers, specified in procurement contracts, is the standard for leading pharmaceutical companies. Green chemistry metrics — specifically Process Mass Intensity — should be included in API supplier scorecards as standard practice. EU supply chain due diligence laws impose legal compliance obligations that must be translated into supplier audit requirements and procurement contract terms.
8. Investment Strategy for Pharmaceutical Procurement Intelligence
8.1 Using Patent Expiration Data for Portfolio Positioning
The pharmaceutical patent cliff is one of the most predictable structural events in equity markets. When a drug’s composition of matter patent expires and the first generic or biosimilar enters, brand revenue typically falls 40-80% within 12 months. This cliff is visible years in advance through Orange Book patent data, Paragraph IV filing records, and ANDA approval databases. Investors who track this data can position ahead of the inflection with precision that fundamentals-only analysis cannot match.
The key data inputs are composition of matter patent expiration (including PTE adjustments), the Paragraph IV certification pipeline (which provides advance notice of competitive intent), the ANDA approval queue (which indicates how many competing generics are ready to launch simultaneously), and any authorized generic agreements (which the brand company may pre-negotiate to capture a portion of the generic market and suppress the first-to-file exclusivity value).
Brand erosion speed is determined by the number of generic entrants at launch, the presence or absence of an authorized generic, and whether any of the generics carry interchangeable designation (for biologics). A brand drug with one generic entrant (180-day exclusivity) retains far more revenue share than a brand drug with eight generics entering simultaneously, because the latter triggers immediate commoditization.
8.2 CDMO Capacity as a Leading Indicator
CDMO contract wins are visible through several data sources before they appear in financial disclosures. Drug Master File submissions to the FDA indicate which manufacturers are activating new customer relationships for specific APIs. Manufacturing supplement filings (Prior Approval Supplements, CBE-30s) indicate when a drug product manufacturer is adding a new contract manufacturing site. Clinical trial IND filings cross-referenced with CDMO public statements can identify early-stage manufacturing relationships.
Investors tracking CDMO capacity utilization — particularly in biologics fill-finish, which is the most constrained segment of the biologics supply chain — can anticipate pricing power inflections ahead of earnings calls. Lyophilization capacity, a critical capability for biologic and peptide drug products, is in chronic shortage globally; CDMOs that have announced lyophilizer capacity expansions represent near-term beneficiaries of the backlog.
8.3 Procurement as a Signal for M&A Activity
Large pharmaceutical companies acquire smaller biotechs partly to access their IP portfolios — but also to bring their drug pipelines in-house for manufacturing and procurement. An acquirer’s procurement function evaluates the target’s supply chain before closing: the CMO relationships, the API sourcing arrangements, and the regulatory compliance status of each manufacturing site. Supply chain deficiencies identified in due diligence regularly reduce deal valuations or introduce escrow holdbacks.
For investors, a biotech with a well-documented, audit-ready supply chain — including validated CMO relationships, FDA-inspected API suppliers, and current Quality Agreements — commands a structurally higher acquisition multiple than a comparable biotech with underdeveloped supply chain infrastructure. Supply chain diligence quality is now explicitly addressed in acquisition term sheets, making procurement capability a disclosed valuation driver in the M&A process.
9. Comprehensive FAQ
Q: What is the difference between a Type II DMF and a CEP for an API supplier qualification?
A: A Type II Drug Master File (DMF) is a confidential submission to the FDA by an API manufacturer that allows the FDA to review manufacturing process, specification, and analytical data in support of a drug product application (NDA, ANDA, BLA) without disclosing that information to the applicant. A Certificate of Suitability (CEP) is issued by the European Directorate for the Quality of Medicines (EDQM) to certify that an API’s quality specification is controlled by a monograph in the European Pharmacopoeia. The CEP is the European equivalent of the DMF for European regulatory filings and is broadly accepted by regulatory authorities in countries that participate in the EDQM certification system. In the U.S., only a Type II DMF supports FDA review; a CEP alone is insufficient.
Q: How does the DSCSA impact pharmaceutical procurement contracts?
A: DSCSA requires that all trading partners be Authorized Trading Partners. Procurement contracts must mandate annual licensure verification, EPCIS-compliant serialized data exchange for every shipment, and supplier cooperation in suspect product investigations. Contracts should specify resolution SLAs for ‘Data, No Product’ exceptions (typically 24-48 hours), assign the cost of quarantine and demurrage to the supplier in the event of data failures, and provide the buyer with the right to return product and cancel purchase orders if exceptions are not resolved within the SLA window.
Q: What is a ‘skinny label’ and why does it create procurement risk for distributors and health systems?
A: A skinny label (Section viii carve-out) is a generic drug label that omits patent-protected indications to allow earlier market entry. It creates procurement risk for buyers because courts have found that induced infringement liability can attach to anyone in the distribution chain who actively encourages use of the generic for the patented indication. Procurement contracts for skinny label products must include manufacturer indemnification against patent infringement claims and promotional restriction clauses prohibiting marketing materials that reference the carved-out indication.
Q: How does the BIOSECURE Act affect API sourcing strategies for companies with NIH funding?
A: The BIOSECURE Act restricts federal grantees from using the equipment or services of designated biotechnology companies of concern. Companies with NIH funding must audit their supply chains, including CRO and CDMO partner facilities, to identify any use of designated entity equipment or services. The ‘equipment or services’ language is broad enough to cover genetic sequencing platforms installed at third-party research facilities. Technology transfer away from designated CDMOs requires 36-60 months for complex biologics; transition planning must begin immediately for companies targeting 2032 compliance.
Q: What is the difference between a Quality Agreement and a Supply Agreement?
A: A Supply Agreement covers commercial terms — price, volume commitments, delivery schedules, liability caps, payment terms, and termination provisions. A Quality Agreement covers GMP responsibilities — batch release authority, deviation investigation ownership, change control notification requirements, audit rights, and complaint handling. They must be separate documents. The Quality Agreement must contain no commercial terms, ensuring that quality decisions (including batch rejection) are made on regulatory grounds without financial pressure. This separation is an FDA expectation and an EMA requirement under Annex 16.
Q: Why is Scope 3 emissions reporting becoming a procurement requirement rather than a voluntary initiative?
A: EU corporate sustainability disclosure rules (CSRD) and supply chain due diligence laws (CSDDD, LkSG) create legal obligations for Scope 3 reporting and supplier environmental due diligence for companies operating in or with significant EU exposure. In the U.S., the SEC’s climate disclosure rules (still subject to legal challenge as of early 2026) proposed mandatory Scope 3 reporting for large accelerated filers. Independently of regulation, pharmaceutical companies have committed to net zero targets that are mathematically impossible without Scope 3 reduction — and Scope 3 Category 1 (purchased goods and services) is the largest single contributor to the total footprint. Procurement contracts that do not require primary emissions data and supplier decarbonization commitments are incompatible with published corporate net zero targets.
Q: How can a procurement team quantify the financial risk of holding a supplier with FDA enforcement history?
A: Build a risk-weighted procurement model. Assign each supplier a probability of supply disruption (P) based on the severity and recency of its FDA enforcement history — Warning Letters carry higher P than Form 483 observations alone; Import Alerts carry the highest P because they actively block product entry. Then calculate the Expected Loss (EL) as P multiplied by the financial impact of a supply disruption for that API: the cost of an alternative source (if one exists), the revenue at risk from production delays, and the cost of an emergency regulatory filing to qualify an alternative site. Suppliers with high EL should carry a risk premium in the TCO model that can justify higher prices for more reliable domestic alternatives.
This analysis draws on FDA Orange Book data, DSCSA regulatory text, the enacted BIOSECURE Act, USP Medicine Supply Map methodology, GHG Protocol Scope 3 Category 1 guidance, and DrugPatentWatch patent and litigation tracking databases. Data points should be verified against primary sources before use in investment decisions or procurement strategy.