DOXYCYCLINE HYCLATE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Doxycycline Hyclate, and when can generic versions of Doxycycline Hyclate launch?
Doxycycline Hyclate is a drug marketed by Pliva, Bausch, Actavis Labs Fl Inc, Ajanta Pharma Ltd, Alembic, Amneal Pharms, Changzhou Pharm, Chartwell, Halsey, Heather, Hikma Intl Pharms, Interpharm, Mutual Pharm, Nesher Pharms, Nostrum Labs Inc, Par Pharm, Pvt Form, Ranbaxy, Strides Pharma, Sun Pharm Industries, Superpharm, Warner Chilcott, Watson Labs, Zhejiang Yongtai, Zydus Lifesciences, Gland Pharma Ltd, Par Sterile Products, West-ward Pharms Int, Actavis Elizabeth, Aurobindo Pharma Usa, Heritage Pharms, Impax Labs Inc, Lupin, Prinston Inc, Rising, Zydus Pharms, Acella, Amneal, Amneal Pharms Co, Apotex, Avet Lifesciences, Caribe Holdings, Chartwell Molecular, Dr Reddys Labs Sa, Epic Pharma Llc, Heritage Pharma, Larken Labs, Lupin Ltd, Mylan, Novel Labs Inc, Pharmobedient, and Praxgen. and is included in seventy-six NDAs.
The generic ingredient in DOXYCYCLINE HYCLATE is doxycycline hyclate. There are twenty-eight drug master file entries for this compound. Seventy-two suppliers are listed for this compound. Additional details are available on the doxycycline hyclate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Doxycycline Hyclate
A generic version of DOXYCYCLINE HYCLATE was approved as doxycycline hyclate by CARIBE HOLDINGS on November 8th, 1982.
Summary for DOXYCYCLINE HYCLATE
| US Patents: | 0 |
| Applicants: | 52 |
| NDAs: | 76 |
| Finished Product Suppliers / Packagers: | 64 |
| Raw Ingredient (Bulk) Api Vendors: | 194 |
| Clinical Trials: | 24 |
| Patent Applications: | 4,097 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for DOXYCYCLINE HYCLATE |
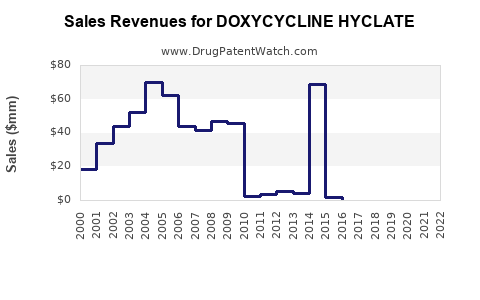
| Drug Sales Revenues: | Drug sales revenues for DOXYCYCLINE HYCLATE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DOXYCYCLINE HYCLATE |
| What excipients (inactive ingredients) are in DOXYCYCLINE HYCLATE? | DOXYCYCLINE HYCLATE excipients list |
| DailyMed Link: | DOXYCYCLINE HYCLATE at DailyMed |
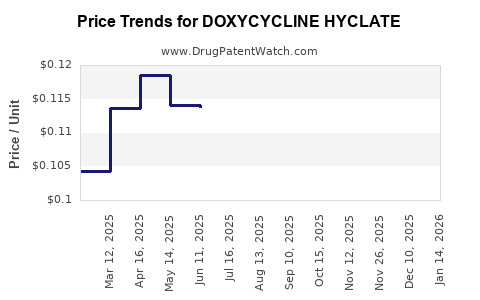
See drug prices for DOXYCYCLINE HYCLATE
Recent Clinical Trials for DOXYCYCLINE HYCLATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Zhuzhou Qianjin Pharmaceutical Co., Ltd., 801, Zhuzhou, China | Phase 1/Phase 2 |
| The Jinnah Postgraduate Medical Centre, Rafiqui، Sarwar Shaheed Rd, Karachi Cantonment, Karachi, Karachi City, Sindh 75510 | Phase 1/Phase 2 |
| University of Karachi | Phase 1/Phase 2 |
Pharmacology for DOXYCYCLINE HYCLATE
| Drug Class | Tetracycline-class Drug |
Anatomical Therapeutic Chemical (ATC) Classes for DOXYCYCLINE HYCLATE
Paragraph IV (Patent) Challenges for DOXYCYCLINE HYCLATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| DORYX MPC | Delayed-release Tablets | doxycycline hyclate | 60 mg and 120 mg | 050795 | 1 | 2017-09-28 |
| DORYX MPC | Delayed-release Tablets | doxycycline hyclate | 50 mg | 050795 | 1 | 2015-11-05 |
| DORYX MPC | Delayed-release Tablets | doxycycline hyclate | 80 mg | 050795 | 1 | 2015-07-01 |
| DORYX MPC | Delayed-release Tablets | doxycycline hyclate | 200 mg | 050795 | 1 | 2014-05-19 |
| DORYX MPC | Delayed-release Tablets | doxycycline hyclate | 150 mg | 050795 | 1 | 2008-12-19 |
US Patents and Regulatory Information for DOXYCYCLINE HYCLATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin | DOXYCYCLINE HYCLATE | doxycycline hyclate | TABLET, DELAYED RELEASE;ORAL | 208741-003 | Aug 11, 2023 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Apotex | DOXYCYCLINE HYCLATE | doxycycline hyclate | TABLET;ORAL | 209243-001 | Apr 15, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Warner Chilcott | DOXYCYCLINE HYCLATE | doxycycline hyclate | CAPSULE;ORAL | 062594-001 | Dec 5, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


