Lupin Ltd Company Profile
✉ Email this page to a colleague
What is the competitive landscape for LUPIN LTD, and what generic alternatives to LUPIN LTD drugs are available?
LUPIN LTD has one hundred and fifty-eight approved drugs.
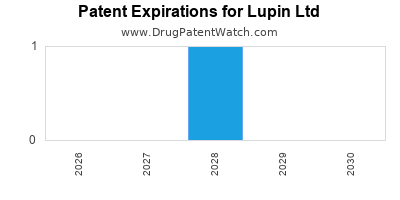
There is one US patent protecting LUPIN LTD drugs. There are nineteen tentative approvals on LUPIN LTD drugs.
There are two patent family members on LUPIN LTD drugs in two countries and five hundred and eighty-three supplementary protection certificates in seventeen countries.
Summary for Lupin Ltd
| International Patents: | 2 |
| US Patents: | 1 |
| Tradenames: | 137 |
| Ingredients: | 120 |
| NDAs: | 158 |
| Drug Master File Entries: | 194 |
| Patent Litigation for Lupin Ltd: | See patent lawsuits for Lupin Ltd |
| PTAB Cases with Lupin Ltd as petitioner: | See PTAB cases with Lupin Ltd as petitioner |
Drugs and US Patents for Lupin Ltd
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin Ltd | IMIPRAMINE PAMOATE | imipramine pamoate | CAPSULE;ORAL | 090444-001 | Apr 16, 2010 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | IMIPRAMINE PAMOATE | imipramine pamoate | CAPSULE;ORAL | 090444-003 | Apr 16, 2010 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | BUPROPION HYDROCHLORIDE | bupropion hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 090693-002 | Apr 6, 2017 | AB3 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | DULOXETINE HYDROCHLORIDE | duloxetine hydrochloride | CAPSULE, DELAYED REL PELLETS;ORAL | 090694-004 | Dec 11, 2013 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | FEBUXOSTAT | febuxostat | TABLET;ORAL | 205406-002 | Jan 17, 2024 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | SUPRAX | cefixime | TABLET, CHEWABLE;ORAL | 065380-002 | Oct 25, 2010 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Paragraph IV (Patent) Challenges for LUPIN LTD drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | for Oral Suspension | 500 mg/5 mL | ➤ Subscribe | 2014-07-22 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
International Patents for Lupin Ltd Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2007119249 | ⤷ Try a Trial |
| Germany | 112007000920 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Lupin Ltd Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2957286 | 132019000000021 | Italy | ⤷ Try a Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM E QUALSIASI SUO SALE O DERIVATO(VELTASSA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/17/1179, 20170721 |
| 0277829 | SPC/GB00/024 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: GANIRELIX OR PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTERED: UK EU/1/00/130/001 20000517; UK EU/1/00/130/002 20000517 |
| 0277829 | C300016 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: GANIRELIX, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; NATL REGISTRATION NO/DATE: EU/1/00/130/001-002 20000517 |
| 1412357 | PA2008013,C1412357 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: SITAGLIPTINUM PHOSPHAS MONOHYDRICUS, METFORMINI HYDROCHLORIDUM; REGISTRATION NO/DATE: EU/1/08/455/001 - EU/1/08/455/014 20080716 |
| 0136011 | 2000C/027 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOLUM / NORETHISTERONI ACETAS; NAT. REGISTRATION NO/DATE: 19 IS 106 F3 20000911; FIRST REGISTRATION: NL RVG 23909 19991124 |
| 0660716 | C300099 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BIMATOPROST; NAT. REGISTRATION NO/DATE: EU/1/02/205/001 20020308; FIRST REGISTRATION: EU/1/02/205/001 20020308 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.