HYDROCODONE Drug Patent Profile
✉ Email this page to a colleague
When do Hydrocodone patents expire, and what generic alternatives are available?
Hydrocodone is a drug marketed by Nostrum Labs Inc, Alvogen, Mallinckrodt, Mikart, Chartwell, Genus, Mallinckrodt Inc, Nesher Pharms, Pharm Assoc, Tris Pharma Inc, Vintage Pharms, Wes Pharma Inc, Abhai Llc, Able, Actavis Labs Fl Inc, Amneal Pharms, Amneal Pharms Ny, Apil, Ascent Pharms Inc, Aurolife Pharma Llc, Barr, Caraco, Cerovene Inc, Epic Pharma Llc, Granules, Halsey, Ivax Pharms, Lannett Co Inc, Mutual Pharm, Novel Labs Inc, Prinston Inc, Ranbaxy, Ranbaxy Labs Ltd, Rhodes Pharms, Sandoz, Specgx Llc, Strides Pharma, Sun Pharm Inds Inc, Sun Pharm Inds Ltd, Ucb Inc, Upsher Smith Labs, Usl Pharma, Vintage Pharms Llc, Watson Labs, Watson Labs Florida, Lgm Pharma, Acella, Actavis Mid Atlantic, Apozeal Pharms, Ivax Sub Teva Pharms, Padagis Us, Pai Holdings Pharm, Wockhardt Bio Ag, Ani Pharms, Teva, Mayne Pharma Inc, and Neos Theraps Inc. and is included in one hundred and fifty-seven NDAs.
The generic ingredient in HYDROCODONE is chlorpheniramine polistirex; hydrocodone polistirex. There are twenty-nine drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the chlorpheniramine polistirex; hydrocodone polistirex profile page.
Summary for HYDROCODONE
| US Patents: | 0 |
| Applicants: | 57 |
| NDAs: | 157 |
| Raw Ingredient (Bulk) Api Vendors: | 19 |
| Clinical Trials: | 135 |
| Patent Applications: | 3,926 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for HYDROCODONE |
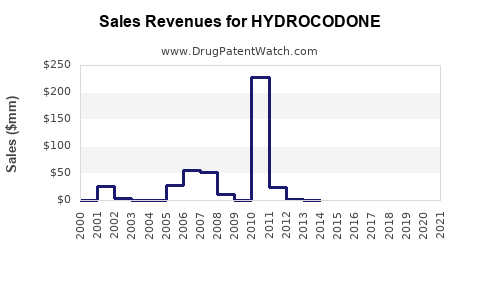
| Drug Sales Revenues: | Drug sales revenues for HYDROCODONE |
| DailyMed Link: | HYDROCODONE at DailyMed |
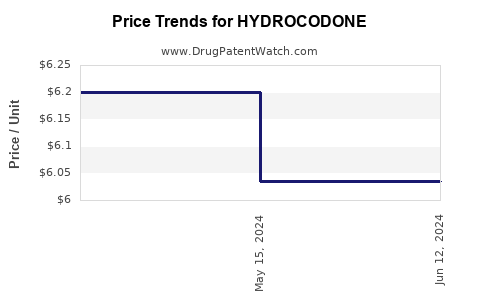
See drug prices for HYDROCODONE
Recent Clinical Trials for HYDROCODONE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Institute on Drug Abuse (NIDA) | Phase 1 |
| Elysium Therapeutics, Inc. | Phase 1 |
| Medpace, Inc. | Phase 1 |
Anatomical Therapeutic Chemical (ATC) Classes for HYDROCODONE
US Patents and Regulatory Information for HYDROCODONE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amneal Pharms Ny | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 040754-001 | Aug 25, 2006 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Wes Pharma Inc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 210211-003 | Oct 30, 2017 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Cerovene Inc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 211690-001 | Feb 7, 2020 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Actavis Labs Fl Inc | HYDROCODONE BITARTRATE AND IBUPROFEN | hydrocodone bitartrate; ibuprofen | TABLET;ORAL | 076604-001 | Dec 31, 2003 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Abhai Llc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 209036-003 | Jun 21, 2017 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Pharm Assoc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | SOLUTION;ORAL | 040838-001 | May 10, 2013 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Specgx Llc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 206718-003 | Mar 31, 2017 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


