Tris Pharma Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TRIS PHARMA INC, and when can generic versions of TRIS PHARMA INC drugs launch?
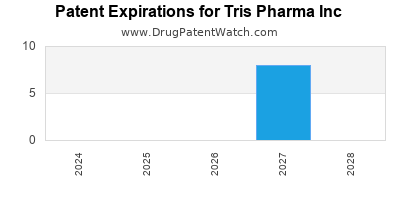
TRIS PHARMA INC has twenty-eight approved drugs.
There are eleven US patents protecting TRIS PHARMA INC drugs.
There are nineteen patent family members on TRIS PHARMA INC drugs in fourteen countries and twenty-eight supplementary protection certificates in ten countries.
Summary for Tris Pharma Inc
| International Patents: | 19 |
| US Patents: | 11 |
| Tradenames: | 28 |
| Ingredients: | 23 |
| NDAs: | 28 |
| Drug Master File Entries: | 6 |
| Patent Litigation for Tris Pharma Inc: | See patent lawsuits for Tris Pharma Inc |
Drugs and US Patents for Tris Pharma Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tris Pharma Inc | HYDROCODONE POLISTIREX AND CHLORPHENIRAMINE POLISTIREX | chlorpheniramine polistirex; hydrocodone polistirex | SUSPENSION, EXTENDED RELEASE;ORAL | 091632-001 | Oct 1, 2010 | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Tris Pharma Inc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 202214-006 | Mar 15, 2016 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Tris Pharma Inc | DEXMETHYLPHENIDATE HYDROCHLORIDE | dexmethylphenidate hydrochloride | TABLET;ORAL | 207901-002 | Aug 26, 2016 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Tris Pharma Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2396039 | ⤷ Try a Trial |
| China | 101400343 | ⤷ Try a Trial |
| Canada | 2645855 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Tris Pharma Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1718641 | 2012/008 | Ireland | ⤷ Try a Trial | PRODUCT NAME: AZILSARTAN MEDOXOMIL AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF, INCLUDING THE POTASSIUM SALT; REGISTRATION NO/DATE: EU/1/11/734/001-011 EU/1/11/735/001-011 20111209 |
| 1441735 | 08C0026 | France | ⤷ Try a Trial | PRODUCT NAME: RALTEGRAVIR POTASSIUM; REGISTRATION NO/DATE: EU/1/07/436/001 20080102 |
| 1539166 | 92323 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: LA COMBINAISON DE (A) DEXTROMETHORPHAN OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES, PRECURSEURS OU DERIVES , PAR EX. HYDROBROMURE DE DEXTROMETHORPHAN ET EN PARTICULIER HYDROBROMURE DE DEXTROMETHORPHAN MONOHYDRATE; ET(B) QUINIDINE OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES , PRECURSEURS OU DERIVES, PAR EX. SULFATE DE QUINIDINE ET EN PARTICULIER SULFATE DE QUINIDINE DEHYDRATE; FIRST REGISTRATION: 20130624 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.