Spain: These 52 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Spain
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Spain: These 52 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025" DrugPatentWatch.com thinkBiotech, 2024 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: Spain Patent 2,374,553
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Spain Patent 2,360,811
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Spain Patent 2,408,216
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Spain Patent 2,510,840
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Spain Patent 2,549,763
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can ARCAPTA NEOHALER (indacaterol maleate) generic drug versions launch?
Generic name: indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Spain Patent 2,362,429

This drug has eighty-six patent family members in thirty-nine countries.
See drug price trends for ARCAPTA NEOHALER.
The generic ingredient in ARCAPTA NEOHALER is indacaterol maleate. Additional details are available on the indacaterol maleate profile page.
When can UTIBRON (glycopyrrolate ; indacaterol maleate) generic drug versions launch?
Generic name: glycopyrrolate ; indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Spain Patent 2,362,429
UTIBRON is a drug marketed by Novartis. There are three patents protecting this drug.
This drug has one hundred and forty-eight patent family members in forty countries. There has been litigation on patents covering UTIBRON
See drug price trends for UTIBRON.
The generic ingredient in UTIBRON is glycopyrrolate ; indacaterol maleate. There are seventeen drug master file entries for this API. Additional details are available on the glycopyrrolate ; indacaterol maleate profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: Spain Patent 2,388,441
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: Spain Patent 2,960,824
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can CAPRELSA (vandetanib) generic drug versions launch?
Generic name: vandetanib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: Spain Patent 2,330,025
CAPRELSA is a drug marketed by Genzyme Corp. There is one patent protecting this drug.
This drug has forty patent family members in thirty-three countries.
See drug price trends for CAPRELSA.
The generic ingredient in CAPRELSA is vandetanib. One supplier is listed for this generic product. Additional details are available on the vandetanib profile page.
When can QNASL (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: Spain Patent 2,339,574

This drug has fifty-two patent family members in thirty-two countries.
See drug price trends for QNASL.
The generic ingredient in QNASL is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,582,646

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,593,582

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,754,573

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,582,646

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,593,582

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Spain Patent 2,754,573

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can FOLOTYN (pralatrexate) generic drug versions launch?
Generic name: pralatrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 30, 2024
Generic Entry Controlled by: Spain Patent 2,313,365
FOLOTYN is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-one patent family members in twenty-three countries. There has been litigation on patents covering FOLOTYN
See drug price trends for FOLOTYN.
The generic ingredient in FOLOTYN is pralatrexate. There are two drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pralatrexate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Spain Patent 2,748,200
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: Spain Patent 2,303,255
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the rifamycin profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,291,780

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,359,504

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,291,780

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,359,504

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,361,325
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,564,185
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Spain Patent 2,627,917
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Spain Patent 2,404,697

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can ARNUITY ELLIPTA (fluticasone furoate) generic drug versions launch?
Generic name: fluticasone furoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2024
Generic Entry Controlled by: Spain Patent 2,561,421
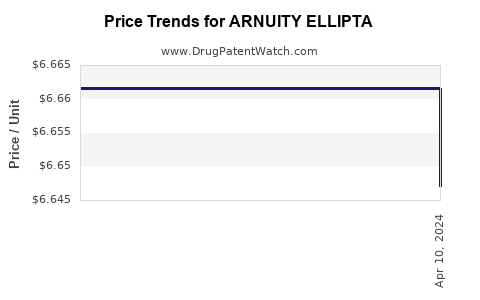
This drug has one hundred and seventeen patent family members in twenty-seven countries. There has been litigation on patents covering ARNUITY ELLIPTA
See drug price trends for ARNUITY ELLIPTA.
The generic ingredient in ARNUITY ELLIPTA is fluticasone furoate. There are twenty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the fluticasone furoate profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: Spain Patent 2,377,430
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Spain Patent 2,529,574
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: Spain Patent 2,901,955

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can YONDELIS (trabectedin) generic drug versions launch?
Generic name: trabectedin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: Spain Patent 2,290,844
YONDELIS is a drug marketed by Janssen Prods. There is one patent protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has forty-four patent family members in thirty-six countries. There has been litigation on patents covering YONDELIS
See drug price trends for YONDELIS.
The generic ingredient in YONDELIS is trabectedin. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the trabectedin profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: Spain Patent 2,456,946
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: Spain Patent 2,951,567
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 02, 2024
Generic Entry Controlled by: Spain Patent 2,348,249

This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the nepafenac profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Spain Patent 2,370,136
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Spain Patent 2,375,788
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.
When can ISENTRESS (raltegravir potassium) generic drug versions launch?
Generic name: raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Spain Patent 2,370,136

This drug has one hundred and twenty-five patent family members in forty-five countries.
See drug price trends for ISENTRESS.
The generic ingredient in ISENTRESS is raltegravir potassium. There are five drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the raltegravir potassium profile page.
When can ISENTRESS (raltegravir potassium) generic drug versions launch?
Generic name: raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Spain Patent 2,375,788

This drug has one hundred and twenty-five patent family members in forty-five countries.
See drug price trends for ISENTRESS.
The generic ingredient in ISENTRESS is raltegravir potassium. There are five drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the raltegravir potassium profile page.
When can NEURACEQ (florbetaben f-18) generic drug versions launch?
Generic name: florbetaben f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 17, 2024
Generic Entry Controlled by: Spain Patent 2,526,655
NEURACEQ is a drug marketed by Life Molecular. There is one patent protecting this drug.
This drug has thirty-five patent family members in twenty-seven countries.
The generic ingredient in NEURACEQ is florbetaben f-18. One supplier is listed for this generic product. Additional details are available on the florbetaben f-18 profile page.
When can BELVIQ (lorcaserin hydrochloride) generic drug versions launch?
Generic name: lorcaserin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 21, 2024
Generic Entry Controlled by: Spain Patent 2,332,009

This drug has thirty-five patent family members in twenty-seven countries. There has been litigation on patents covering BELVIQ
See drug price trends for BELVIQ.
The generic ingredient in BELVIQ is lorcaserin hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the lorcaserin hydrochloride profile page.
When can RECORLEV (levoketoconazole) generic drug versions launch?
Generic name: levoketoconazole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 10, 2025
Generic Entry Controlled by: Spain Patent 2,377,526
RECORLEV is a drug marketed by Strongbridge. There are seven patents protecting this drug.
This drug has thirty-two patent family members in twenty countries.
See drug price trends for RECORLEV.
The generic ingredient in RECORLEV is levoketoconazole. One supplier is listed for this generic product. Additional details are available on the levoketoconazole profile page.
When can BELRAPZO (bendamustine hydrochloride) generic drug versions launch?
Generic name: bendamustine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 14, 2025
Generic Entry Controlled by: Spain Patent 2,405,611
BELRAPZO is a drug marketed by Eagle Pharms. There are nine patents protecting this drug. Six tentatively approved generics are ready to enter the market.
This drug has sixty-four patent family members in thirty countries.
See drug price trends for BELRAPZO.
The generic ingredient in BELRAPZO is bendamustine hydrochloride. There are twenty-three drug master file entries for this API. Ten suppliers are listed for this generic product. Additional details are available on the bendamustine hydrochloride profile page.
When can BENDEKA (bendamustine hydrochloride) generic drug versions launch?
Generic name: bendamustine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 14, 2025
Generic Entry Controlled by: Spain Patent 2,405,611
BENDEKA is a drug marketed by Eagle Pharms. There are eighteen patents protecting this drug and one Paragraph IV challenge. Six tentatively approved generics are ready to enter the market.
This drug has one hundred and fourteen patent family members in thirty-one countries. There has been litigation on patents covering BENDEKA
See drug price trends for BENDEKA.
The generic ingredient in BENDEKA is bendamustine hydrochloride. There are twenty-three drug master file entries for this API. Ten suppliers are listed for this generic product. Additional details are available on the bendamustine hydrochloride profile page.
When can OTREXUP (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 24, 2025
Generic Entry Controlled by: Spain Patent 2,716,135

This drug has thirty-seven patent family members in fourteen countries. There has been litigation on patents covering OTREXUP
See drug price trends for OTREXUP.
The generic ingredient in OTREXUP is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can OTREXUP (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 24, 2025
Generic Entry Controlled by: Spain Patent 2,908,027

This drug has thirty-seven patent family members in fourteen countries. There has been litigation on patents covering OTREXUP
See drug price trends for OTREXUP.
The generic ingredient in OTREXUP is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can WAKIX (pitolisant hydrochloride) generic drug versions launch?
Generic name: pitolisant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 10, 2025
Generic Entry Controlled by: Spain Patent 2,309,948
WAKIX is a drug marketed by Harmony. There are three patents protecting this drug.
This drug has sixty-one patent family members in thirty-one countries. There has been litigation on patents covering WAKIX
See drug price trends for WAKIX.
The generic ingredient in WAKIX is pitolisant hydrochloride. One supplier is listed for this generic product. Additional details are available on the pitolisant hydrochloride profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,414,095

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,540,966

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,686,981

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,872,962

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,414,095

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering KERYDIN
See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,540,966

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering KERYDIN
See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,686,981

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering KERYDIN
See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 16, 2025
Generic Entry Controlled by: Spain Patent 2,872,962

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering KERYDIN
See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,437,268
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,446,324
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,567,197
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,569,357
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,667,868
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,743,531
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,892,304
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 21, 2025
Generic Entry Controlled by: Spain Patent 2,906,792
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-five patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can CORLANOR (ivabradine) generic drug versions launch?
Generic name: ivabradine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 28, 2025
Generic Entry Controlled by: Spain Patent 2,308,689

This drug has ninety-seven patent family members in forty-two countries. There has been litigation on patents covering CORLANOR
See drug price trends for CORLANOR.
The generic ingredient in CORLANOR is ivabradine. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivabradine profile page.
When can CORLANOR (ivabradine hydrochloride) generic drug versions launch?
Generic name: ivabradine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 28, 2025
Generic Entry Controlled by: Spain Patent 2,313,581

This drug has ninety-seven patent family members in forty-two countries. There has been litigation on patents covering CORLANOR
See drug price trends for CORLANOR.
The generic ingredient in CORLANOR is ivabradine hydrochloride. There are nine drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the ivabradine hydrochloride profile page.
When can XIFAXAN (rifaximin) generic drug versions launch?
Generic name: rifaximin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 03, 2025
Generic Entry Controlled by: Spain Patent 2,522,895
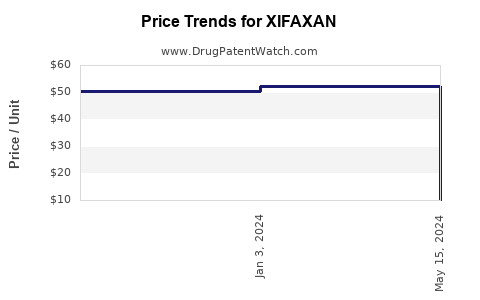
This drug has two hundred and eleven patent family members in forty countries. There has been litigation on patents covering XIFAXAN
See drug price trends for XIFAXAN.
The generic ingredient in XIFAXAN is rifaximin. There are fourteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the rifaximin profile page.
When can NEXAVAR (sorafenib tosylate) generic drug versions launch?
Generic name: sorafenib tosylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 07, 2025
Generic Entry Controlled by: Spain Patent 2,351,612
NEXAVAR is a drug marketed by Bayer Hlthcare. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has eighty-nine patent family members in thirty-nine countries. There has been litigation on patents covering NEXAVAR
See drug price trends for NEXAVAR.
The generic ingredient in NEXAVAR is sorafenib tosylate. There are thirteen drug master file entries for this API. Eight suppliers are listed for this generic product. Additional details are available on the sorafenib tosylate profile page.
When can VALCHLOR (mechlorethamine hydrochloride) generic drug versions launch?
Generic name: mechlorethamine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 14, 2025
Generic Entry Controlled by: Spain Patent 2,388,744
VALCHLOR is a drug marketed by Helsinn. There are six patents protecting this drug.
This drug has fifty patent family members in twenty countries.
See drug price trends for VALCHLOR.
The generic ingredient in VALCHLOR is mechlorethamine hydrochloride. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the mechlorethamine hydrochloride profile page.
When can VALCHLOR (mechlorethamine hydrochloride) generic drug versions launch?
Generic name: mechlorethamine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 14, 2025
Generic Entry Controlled by: Spain Patent 2,727,523
VALCHLOR is a drug marketed by Helsinn. There are six patents protecting this drug.
This drug has fifty patent family members in twenty countries.
See drug price trends for VALCHLOR.
The generic ingredient in VALCHLOR is mechlorethamine hydrochloride. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the mechlorethamine hydrochloride profile page.
When can VALCHLOR (mechlorethamine hydrochloride) generic drug versions launch?
Generic name: mechlorethamine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 14, 2025
Generic Entry Controlled by: Spain Patent 2,848,841
VALCHLOR is a drug marketed by Helsinn. There are six patents protecting this drug.
This drug has fifty patent family members in twenty countries.
See drug price trends for VALCHLOR.
The generic ingredient in VALCHLOR is mechlorethamine hydrochloride. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the mechlorethamine hydrochloride profile page.
When can OZEMPIC (semaglutide) generic drug versions launch?
Generic name: semaglutide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 18, 2025
Generic Entry Controlled by: Spain Patent 2,350,051

This drug has two hundred and twenty-six patent family members in twenty-eight countries. There has been litigation on patents covering OZEMPIC
See drug price trends for OZEMPIC.
The generic ingredient in OZEMPIC is semaglutide. Two suppliers are listed for this generic product. Additional details are available on the semaglutide profile page.
When can OZEMPIC (semaglutide) generic drug versions launch?
Generic name: semaglutide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 18, 2025
Generic Entry Controlled by: Spain Patent 2,557,313

This drug has two hundred and twenty-six patent family members in twenty-eight countries. There has been litigation on patents covering OZEMPIC
See drug price trends for OZEMPIC.
The generic ingredient in OZEMPIC is semaglutide. Two suppliers are listed for this generic product. Additional details are available on the semaglutide profile page.
When can WEGOVY (semaglutide) generic drug versions launch?
Generic name: semaglutide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 18, 2025
Generic Entry Controlled by: Spain Patent 2,350,051
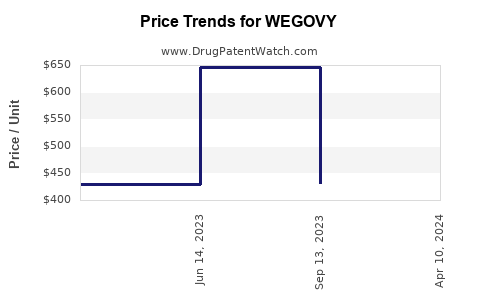
This drug has one hundred and twenty-two patent family members in thirty-two countries. There has been litigation on patents covering WEGOVY
See drug price trends for WEGOVY.
The generic ingredient in WEGOVY is semaglutide. Two suppliers are listed for this generic product. Additional details are available on the semaglutide profile page.
When can WEGOVY (semaglutide) generic drug versions launch?
Generic name: semaglutide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 18, 2025
Generic Entry Controlled by: Spain Patent 2,557,313

This drug has one hundred and twenty-two patent family members in thirty-two countries. There has been litigation on patents covering WEGOVY
See drug price trends for WEGOVY.
The generic ingredient in WEGOVY is semaglutide. Two suppliers are listed for this generic product. Additional details are available on the semaglutide profile page.
When can JATENZO (testosterone undecanoate) generic drug versions launch?
Generic name: testosterone undecanoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 15, 2025
Generic Entry Controlled by: Spain Patent 2,930,658
JATENZO is a drug marketed by Tolmar. There are ten patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has twenty-nine patent family members in fourteen countries. There has been litigation on patents covering JATENZO
See drug price trends for JATENZO.
The generic ingredient in JATENZO is testosterone undecanoate. There are sixty-nine drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the testosterone undecanoate profile page.
When can TYKERB (lapatinib ditosylate) generic drug versions launch?
Generic name: lapatinib ditosylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 19, 2025
Generic Entry Controlled by: Spain Patent 2,601,503
TYKERB is a drug marketed by Novartis. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has twenty-eight patent family members in twenty-six countries.
See drug price trends for TYKERB.
The generic ingredient in TYKERB is lapatinib ditosylate. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the lapatinib ditosylate profile page.
When can XCOPRI (cenobamate) generic drug versions launch?
Generic name: cenobamate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 22, 2025
Generic Entry Controlled by: Spain Patent 2,441,765
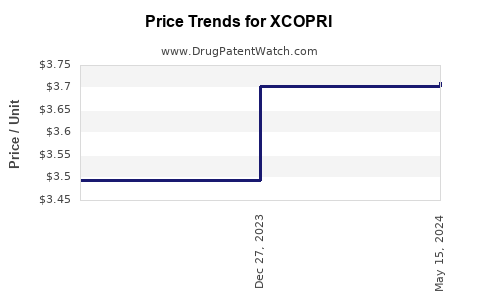
This drug has twenty-six patent family members in twenty countries.
See drug price trends for XCOPRI.
The generic ingredient in XCOPRI is cenobamate. One supplier is listed for this generic product. Additional details are available on the cenobamate profile page.
When can BELEODAQ (belinostat) generic drug versions launch?
Generic name: belinostat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2025
Generic Entry Controlled by: Spain Patent 2,380,887
BELEODAQ is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-nine patent family members in twenty-seven countries. There has been litigation on patents covering BELEODAQ
See drug price trends for BELEODAQ.
The generic ingredient in BELEODAQ is belinostat. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the belinostat profile page.
When can BELEODAQ (belinostat) generic drug versions launch?
Generic name: belinostat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2025
Generic Entry Controlled by: Spain Patent 2,540,204
BELEODAQ is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-nine patent family members in twenty-seven countries. There has been litigation on patents covering BELEODAQ
See drug price trends for BELEODAQ.
The generic ingredient in BELEODAQ is belinostat. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the belinostat profile page.
When can XTANDI (enzalutamide) generic drug versions launch?
Generic name: enzalutamide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2025
Generic Entry Controlled by: Spain Patent 2,378,778
XTANDI is a drug marketed by Astellas. There are four patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and eighty-four patent family members in thirty-four countries. There has been litigation on patents covering XTANDI
See drug price trends for XTANDI.
The generic ingredient in XTANDI is enzalutamide. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the enzalutamide profile page.
When can XTANDI (enzalutamide) generic drug versions launch?
Generic name: enzalutamide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2025
Generic Entry Controlled by: Spain Patent 2,535,179
XTANDI is a drug marketed by Astellas. There are four patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and eighty-four patent family members in thirty-four countries. There has been litigation on patents covering XTANDI
See drug price trends for XTANDI.
The generic ingredient in XTANDI is enzalutamide. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the enzalutamide profile page.
When can TRACLEER (bosentan) generic drug versions launch?
Generic name: bosentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 17, 2025
Generic Entry Controlled by: Spain Patent 2,336,943
TRACLEER is a drug marketed by Actelion. There are two patents protecting this drug and one Paragraph IV challenge. Three tentatively approved generics are ready to enter the market.
This drug has twenty-eight patent family members in twenty-three countries. There has been litigation on patents covering TRACLEER
See drug price trends for TRACLEER.
The generic ingredient in TRACLEER is bosentan. There are nineteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the bosentan profile page.