Harrow Eye Company Profile
✉ Email this page to a colleague
What is the competitive landscape for HARROW EYE, and what generic alternatives to HARROW EYE drugs are available?
HARROW EYE has nineteen approved drugs.
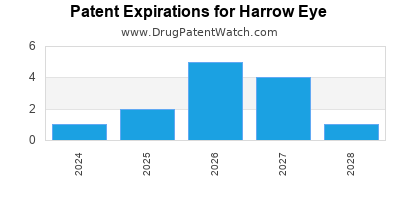
There are twenty-nine US patents protecting HARROW EYE drugs.
There are three hundred and fifty-four patent family members on HARROW EYE drugs in forty-two countries and twenty-five supplementary protection certificates in ten countries.
Drugs and US Patents for Harrow Eye
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Harrow Eye | MAXIDEX | dexamethasone | SUSPENSION/DROPS;OPHTHALMIC | 013422-001 | Approved Prior to Jan 1, 1982 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Harrow Eye | IHEEZO | chloroprocaine hydrochloride | GEL;OPHTHALMIC | 216227-001 | Sep 27, 2022 | RX | Yes | Yes | 10,792,271 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Harrow Eye | VERKAZIA | cyclosporine | EMULSION;OPHTHALMIC | 214965-001 | Jun 23, 2021 | RX | Yes | Yes | 9,132,071 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Harrow Eye
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Harrow Eye | IOPIDINE | apraclonidine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 019779-001 | Dec 31, 1987 | 4,517,199 | ⤷ Try a Trial |
| Harrow Eye | TOBRADEX ST | dexamethasone; tobramycin | SUSPENSION/DROPS;OPHTHALMIC | 050818-001 | Feb 13, 2009 | 5,149,694 | ⤷ Try a Trial |
| Harrow Eye | VIGAMOX | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 021598-001 | Apr 15, 2003 | 4,990,517*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for HARROW EYE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Ophthalmic | 0.50% | ➤ Subscribe | 2005-12-22 |
| ➤ Subscribe | Ophthalmic Solution | 10.5% | ➤ Subscribe | 2012-02-29 |
| ➤ Subscribe | Ophthalmic Suspension | 0.3% | ➤ Subscribe | 2015-12-21 |
International Patents for Harrow Eye Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Denmark | 2262506 | ⤷ Try a Trial |
| Spain | 2910374 | ⤷ Try a Trial |
| Japan | 5894202 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Harrow Eye Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0999825 | CA 2013 00055 | Denmark | ⤷ Try a Trial | PRODUCT NAME: NEPAFENAC (3 MG/ML), HERUNDER NEPAFENAC I KOMBINATION MED GLACTOMANNANPOLYMERER, ISAER 3 MG/ML NEPAFENAC I KOMBINATION MED GALACTOMANNANPOLYMERER, SAMT OFTALMISKE SAMMENSAETNINGER DERAF; REG. NO/DATE: EU1/07/433/002 20130503 |
| 0350733 | 11/2000 | Austria | ⤷ Try a Trial | PRODUCT NAME: MOXIFLOXACIN, DESSEN PHARMAZEUTISCH VERWENDBARE HYDRATE UND SAEUREADDITIONSSALZE, DESSEN ALKALI-ERDALKALI-SILBER- UND GUANIDINIUMSALZE, SOWIE DESSEN C1 - C4 ALKYL- ODER; NAT. REGISTRATION NO/DATE: 1-23494, 1-23495, 1-23496 20000215; FIRST REGISTRATION: DE 45263.00.00 19990621 |
| 1809237 | C01809237/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: CICLOSPORIN; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66093 08.09.2016 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.