Lupin Company Profile
✉ Email this page to a colleague
What is the competitive landscape for LUPIN, and when can generic versions of LUPIN drugs launch?
LUPIN has two hundred and thirty-six approved drugs.
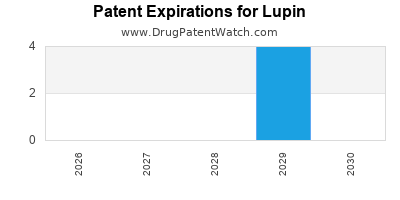
There are nineteen US patents protecting LUPIN drugs. There are twenty-two tentative approvals on LUPIN drugs.
There are ninety-nine patent family members on LUPIN drugs in twenty-four countries and six hundred and eighty-six supplementary protection certificates in seventeen countries.
Summary for Lupin
| International Patents: | 99 |
| US Patents: | 19 |
| Tradenames: | 192 |
| Ingredients: | 172 |
| NDAs: | 236 |
| Patent Litigation for Lupin: | See patent lawsuits for Lupin |
| PTAB Cases with Lupin as petitioner: | See PTAB cases with Lupin as petitioner |
Drugs and US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin Ltd | OLMESARTAN MEDOXOMIL | olmesartan medoxomil | TABLET;ORAL | 206631-003 | Apr 27, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin | SOLOSEC | secnidazole | GRANULE;ORAL | 209363-001 | Sep 15, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin Pharms | MELOXICAM | meloxicam | TABLET;ORAL | 077944-002 | Jul 19, 2006 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin | DOXYCYCLINE HYCLATE | doxycycline hyclate | TABLET, DELAYED RELEASE;ORAL | 208741-004 | Aug 11, 2023 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin Ltd | LEVONORGESTREL | levonorgestrel | TABLET;ORAL | 091328-001 | Jan 23, 2013 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin | SOLOSEC | secnidazole | GRANULE;ORAL | 209363-001 | Sep 15, 2017 | RX | Yes | Yes | 11,000,508 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,844,002 | ⤷ Try a Trial |
| Lupin | ANTARA (MICRONIZED) | fenofibrate | CAPSULE;ORAL | 021695-003 | Nov 30, 2004 | 7,101,574 | ⤷ Try a Trial |
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 7,462,645 | ⤷ Try a Trial |
| Lupin | ANTARA (MICRONIZED) | fenofibrate | CAPSULE;ORAL | 021695-001 | Nov 30, 2004 | 7,101,574 | ⤷ Try a Trial |
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,667,344 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,439,670 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for LUPIN drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | for Oral Suspension | 500 mg/5 mL | ➤ Subscribe | 2014-07-22 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
| ➤ Subscribe | Inhalation Solution | Eq. 0.015 mg base/2 mL | ➤ Subscribe | 2009-10-01 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
International Patents for Lupin Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Cyprus | 1111330 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2004052835 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2013130666 | ⤷ Try a Trial |
| Australia | 2018203882 | ⤷ Try a Trial |
| Eurasian Patent Organization | 201000828 | ⤷ Try a Trial |
| South Korea | 20110010742 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Lupin Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1214076 | 49/2008 | Austria | ⤷ Try a Trial | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON; REGISTRATION NO/DATE: 1-27586 20080612 |
| 0443983 | 98C004 | Belgium | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN HYDROCHLOROTHIAZIDUM; NATL. REGISTRATION NO/DATE: 206 IS 250 F 3 19980624; FIRST REGISTRATION: FR 344300.5 19970925 |
| 2924034 | 122019000042 | Germany | ⤷ Try a Trial | PRODUCT NAME: DORAVIRIN ODER EINEM PHARMAZEUTISCH ANNEHMBAREN SALZ DAVON IN KOMBINATION MIT LAMIVUDIN UND TENOFOVIR-DISOPROXIL-FUMARAT; REGISTRATION NO/DATE: EU/1/18/1333 20181122 |
| 1412357 | DO 77; 5006-2008 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: SITAGLIPTIN A METFORMIN; REGISTRATION NO/DATE: EU/1/08/455/001-014 20080716 |
| 2316456 | 300918 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: NALTREXON OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, IN HET BIJZONDER NALTREXONHYDROCHLORIDE, EN BUPROPION OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, IN HET BIJZONDER BUPROPIONHYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/14/988 20150330 |
| 2435024 | C202130025 | Spain | ⤷ Try a Trial | PRODUCT NAME: UNA COMBINACION DE FORMOTEROL ( INCLUIDAS SUS SALES, ESTERES, SOLVATOS O ENANTIOMEROS FARMACEUTICAMENTE ACEPTABLES I, GLICOPIRROLATO ( INCLUIDAS SUS SALES, ESTERES, SOLVATOS O ENANTIOMEROS FARMACEUTICAMENTE ACEPTABLES ) Y BUDESONIDA ( INCLUIDAS SUS SALES, ESTERES, SOLVATOS O ENANTIOMEROS FARMACEUTICAMENTE ACEPTABLES ).; NATIONAL AUTHORISATION NUMBER: EU/1/20/1498; DATE OF AUTHORISATION: 20201209; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1498; DATE OF FIRST AUTHORISATION IN EEA: 20201209 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.