FUROSEMIDE Drug Patent Profile
✉ Email this page to a colleague
When do Furosemide patents expire, and what generic alternatives are available?
Furosemide is a drug marketed by Abraxis Pharm, Accord Hlthcare, Am Regent, Amneal Pharms Co, Areva Pharms, Astrazeneca, Avet Lifesciences, Baxter Hlthcare Corp, Eugia Pharma, Fresenius Kabi Usa, Gland Pharma Ltd, Hikma, Hospira, Igi Labs Inc, Intl Medication, Mankind Pharma, Marsam Pharms Llc, Meitheal, Sagent, Smith And Nephew, Warner Chilcott, Watson Labs, Wockhardt, Wyeth Ayerst, Pharmobedient Cnsltg, Ani Pharms, Chartwell Rx, Epic Pharma Llc, Graviti Pharms, Ipca Labs Ltd, Kalapharm, Leading, Mylan, Prinston Inc, Strides Pharma, Sun Pharm Inds Inc, Sun Pharm Industries, Superpharm, and Watson Labs Teva. and is included in sixty-four NDAs.
The generic ingredient in FUROSEMIDE is furosemide. There are twenty-two drug master file entries for this compound. Fifty-seven suppliers are listed for this compound. Additional details are available on the furosemide profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Furosemide
A generic version of FUROSEMIDE was approved as furosemide by MYLAN on October 29th, 1986.
Summary for FUROSEMIDE
| US Patents: | 0 |
| Applicants: | 39 |
| NDAs: | 64 |
| Finished Product Suppliers / Packagers: | 55 |
| Raw Ingredient (Bulk) Api Vendors: | 137 |
| Clinical Trials: | 259 |
| Patent Applications: | 3,982 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for FUROSEMIDE |
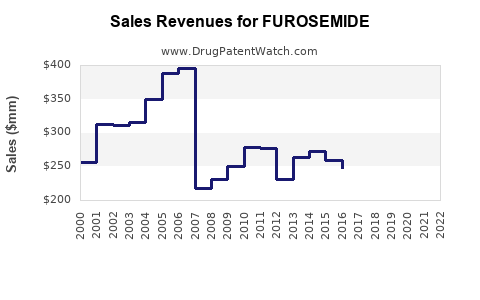
| Drug Sales Revenues: | Drug sales revenues for FUROSEMIDE |
| What excipients (inactive ingredients) are in FUROSEMIDE? | FUROSEMIDE excipients list |
| DailyMed Link: | FUROSEMIDE at DailyMed |
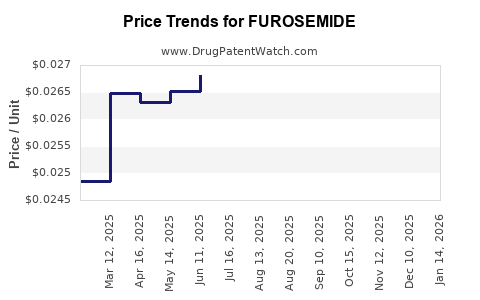
See drug prices for FUROSEMIDE
Recent Clinical Trials for FUROSEMIDE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Children's Hospital Medical Center, Cincinnati | Phase 4 |
| National Heart, Lung, and Blood Institute (NHLBI) | Phase 4 |
| RTI International | Phase 4 |
Pharmacology for FUROSEMIDE
| Drug Class | Loop Diuretic |
| Physiological Effect | Increased Diuresis at Loop of Henle |
Medical Subject Heading (MeSH) Categories for FUROSEMIDE
Anatomical Therapeutic Chemical (ATC) Classes for FUROSEMIDE
US Patents and Regulatory Information for FUROSEMIDE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sun Pharm Inds Inc | FUROSEMIDE | furosemide | TABLET;ORAL | 091258-002 | Apr 1, 2014 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Chartwell Rx | FUROSEMIDE | furosemide | TABLET;ORAL | 018413-002 | Nov 30, 1983 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Wockhardt | FUROSEMIDE | furosemide | INJECTABLE;INJECTION | 077941-001 | Mar 22, 2007 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Watson Labs Teva | FUROSEMIDE | furosemide | TABLET;ORAL | 070449-001 | Nov 22, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Hikma | FUROSEMIDE | furosemide | TABLET;ORAL | 018823-001 | Nov 10, 1983 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Leading | FUROSEMIDE | furosemide | TABLET;ORAL | 077293-002 | Nov 9, 2005 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


