Tetraphase Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TETRAPHASE PHARMS, and what generic alternatives to TETRAPHASE PHARMS drugs are available?
TETRAPHASE PHARMS has one approved drug.
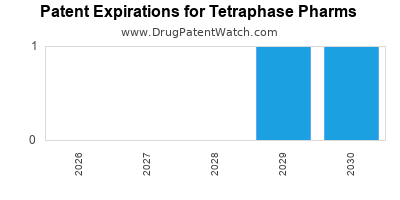
There are four US patents protecting TETRAPHASE PHARMS drugs.
There are seventy-one patent family members on TETRAPHASE PHARMS drugs in thirty-three countries and seventeen supplementary protection certificates in fifteen countries.
Summary for Tetraphase Pharms
| International Patents: | 71 |
| US Patents: | 4 |
| Tradenames: | 1 |
| Ingredients: | 1 |
| NDAs: | 1 |
Drugs and US Patents for Tetraphase Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-001 | Aug 27, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-002 | Jun 3, 2020 | RX | Yes | Yes | 10,961,190 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-002 | Jun 3, 2020 | RX | Yes | Yes | 8,796,245 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-001 | Aug 27, 2018 | RX | Yes | Yes | 8,906,887 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-002 | Jun 3, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-001 | Aug 27, 2018 | RX | Yes | Yes | 11,578,044 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Tetraphase Pharms | XERAVA | eravacycline dihydrochloride | POWDER;INTRAVENOUS | 211109-001 | Aug 27, 2018 | RX | Yes | Yes | 10,961,190 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Tetraphase Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| New Zealand | 603568 | ⤷ Try a Trial |
| Canada | 2732883 | ⤷ Try a Trial |
| Israel | 211120 | ⤷ Try a Trial |
| Serbia | 54485 | ⤷ Try a Trial |
| South Korea | 20110058800 | ⤷ Try a Trial |
| Hong Kong | 1193412 | ⤷ Try a Trial |
| South Korea | 101679023 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Tetraphase Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2323972 | CA 2019 00009 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ERAVACYCLIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/18/1312 20180209 |
| 2323972 | SPC/GB19/017 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ERAVACYCLINE OR PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: UK PLGB 24626/0005 20180924; UK PLGB 24626/0006 20180924; UK EU/1/18/1312 (NI) 20180924 |
| 2323972 | C20190009 00277 | Estonia | ⤷ Try a Trial | PRODUCT NAME: ERAVATSUEKLIIN;REG NO/DATE: EU/1/18/1312 24.09.2018 |
| 2323972 | CR 2019 00009 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ERAVACYCLIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/18/1312 20180209 |
| 2323972 | LUC00107 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ERAVACYCLINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/18/1312 20180924 |
| 2323972 | 19C1012 | France | ⤷ Try a Trial | PRODUCT NAME: ERAVACYCLINE OU L'UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE: EU/1/18/1312 20180924 |
| 2323972 | 715 | Finland | ⤷ Try a Trial | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.