AMNEAL Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AMNEAL, and when can generic versions of AMNEAL drugs launch?
AMNEAL has three hundred and eighty-four approved drugs.
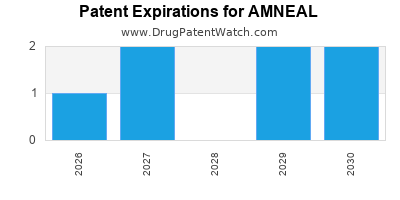
There are five US patents protecting AMNEAL drugs. There are seven tentative approvals on AMNEAL drugs.
There are fifteen patent family members on AMNEAL drugs in twelve countries and eight hundred supplementary protection certificates in eighteen countries.
Summary for AMNEAL
| International Patents: | 15 |
| US Patents: | 5 |
| Tradenames: | 291 |
| Ingredients: | 283 |
| NDAs: | 384 |
| Patent Litigation for AMNEAL: | See patent lawsuits for AMNEAL |
| PTAB Cases with AMNEAL as petitioner: | See PTAB cases with AMNEAL as petitioner |
Drugs and US Patents for AMNEAL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amneal | DOCETAXEL | docetaxel | INJECTABLE;INJECTION | 209640-001 | Jan 19, 2018 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Amneal Pharms | LORAZEPAM | lorazepam | TABLET;ORAL | 078826-002 | Jun 23, 2010 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Amneal | BUPRENORPHINE | buprenorphine | FILM, EXTENDED RELEASE;TRANSDERMAL | 211586-002 | Apr 14, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Amneal | PREDNISONE | prednisone | TABLET;ORAL | 213385-001 | Jun 16, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Amneal Pharms | VALSARTAN AND HYDROCHLOROTHIAZIDE | hydrochlorothiazide; valsartan | TABLET;ORAL | 204382-003 | Aug 11, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Amneal | POTASSIUM PHOSPHATES | potassium phosphate, dibasic; potassium phosphate, monobasic | SOLUTION;INTRAVENOUS | 216344-003 | Oct 10, 2023 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for AMNEAL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Amneal | ZOMIG | zolmitriptan | SPRAY;NASAL | 021450-004 | Sep 30, 2003 | 7,220,767*PED | ⤷ Try a Trial |
| Amneal | ZOMIG | zolmitriptan | SPRAY;NASAL | 021450-004 | Sep 30, 2003 | 5,466,699*PED | ⤷ Try a Trial |
| Amneal | ZOMIG | zolmitriptan | SPRAY;NASAL | 021450-003 | Sep 16, 2013 | 6,750,237*PED | ⤷ Try a Trial |
| Amneal | ZOMIG | zolmitriptan | SPRAY;NASAL | 021450-004 | Sep 30, 2003 | 6,750,237*PED | ⤷ Try a Trial |
| Amneal | ACTIVELLA | estradiol; norethindrone acetate | TABLET;ORAL | 020907-001 | Nov 18, 1998 | RE36247 | ⤷ Try a Trial |
| Amneal | ZOMIG | zolmitriptan | SPRAY;NASAL | 021450-003 | Sep 16, 2013 | 7,220,767*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for AMNEAL drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Nasal Spray | 2.5 mg/spray | ➤ Subscribe | 2016-06-09 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | Injection | 1 mg/mL, 50 mL vials | ➤ Subscribe | 2011-12-16 |
| ➤ Subscribe | Nasal Spray | 5 mg/spray | ➤ Subscribe | 2013-11-14 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Injection | 100 mg/mL, 2.5 mL vials | ➤ Subscribe | 2007-09-24 |
International Patents for AMNEAL Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2013139179 | ⤷ Try a Trial |
| China | 103315947 | ⤷ Try a Trial |
| Portugal | 2881109 | ⤷ Try a Trial |
| Spain | 2795421 | ⤷ Try a Trial |
| Mexico | 2022001139 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2023055457 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for AMNEAL Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0443983 | C00443983/03 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN + AMLODIPINE + HYDROCHLOROTHIAZIDE; REGISTRATION NUMBER/DATE: SWISSMEDIC 59407 16.09.2009 |
| 2101777 | CA 2016 00024 | Denmark | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN USED IN COMBINATION TREATMENT WITH TADALAFIL; REG. NO/DATE: EU/1/08/451 (001-004) 20151125 |
| 0162036 | 2000C/032 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LEVETIRACETAM; NAT. REGISTRATION NO/DATE: EU/1/00/146/001 20000929; FIRST REGISTRATION: CH 55297 20000329 |
| 0633893 | 2012/003 | Ireland | ⤷ Try a Trial | PRODUCT NAME: ZYTIGA (ABIRATERONE) "ABIRATERONE AND ACID ADDITION SALTS AND 3-ESTERS THEREOF, ESPECIALLY ABIRATERONE ACETATE"; REGISTRATION NO/DATE: EU/1/11/714/001 20110905 |
| 1214076 | SZ 49/2008 | Austria | ⤷ Try a Trial | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON |
| 1519731 | C300740 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: AZELASTINE OF EEN; NAT. REGISTRATION NO/DATE: RVG114215 20141125; FIRST REGISTRATION: 2011/07125-REG 20130215 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.