LORATADINE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Loratadine, and when can generic versions of Loratadine launch?
Loratadine is a drug marketed by Aurobindo Pharma, Bionpharma, Marksans Pharma, Strides Pharma, Taro, Aurobindo Pharma Ltd, Hetero Labs Ltd Iii, Lannett Co Inc, Perrigo, Pharm Assoc, Ranbaxy Labs Ltd, Teva, Wockhardt Bio Ag, Perrigo Pharma Intl, Sun Pharm, Actavis Labs Fl Inc, Glaxosmithkline, Rubicon, Tenshi, Apotex Inc, Granules, Guardian Drug, Hetero Labs Ltd V, Mylan, Pld Acquisitions Llc, Sun Pharm Inds Ltd, Unique Pharm, Heritage Pharma, and P And L. and is included in forty-three NDAs.
The generic ingredient in LORATADINE is loratadine; pseudoephedrine sulfate. There are thirty-nine drug master file entries for this compound. Thirty-four suppliers are listed for this compound. Additional details are available on the loratadine; pseudoephedrine sulfate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Loratadine
A generic version of LORATADINE was approved as loratadine; pseudoephedrine sulfate by PERRIGO PHARMA INTL on January 30th, 2003.
Summary for LORATADINE
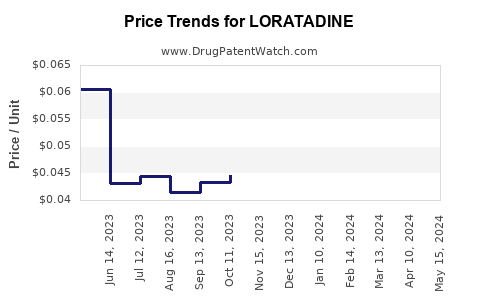
See drug prices for LORATADINE
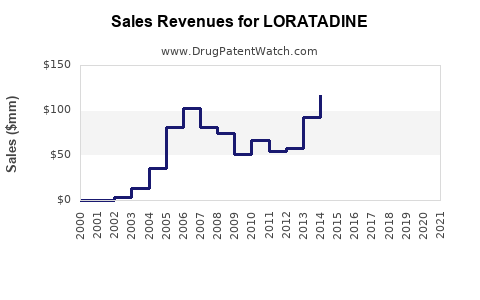
Recent Clinical Trials for LORATADINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| AHS Cancer Control Alberta | Phase 2 |
| King Edward Medical University | Phase 4 |
| Ain Shams University | Phase 3 |
Medical Subject Heading (MeSH) Categories for LORATADINE
Anatomical Therapeutic Chemical (ATC) Classes for LORATADINE
US Patents and Regulatory Information for LORATADINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perrigo Pharma Intl | LORATADINE AND PSEUDOEPHEDRINE SULFATE | loratadine; pseudoephedrine sulfate | TABLET, EXTENDED RELEASE;ORAL | 075989-001 | Mar 4, 2004 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mylan | LORATADINE | loratadine | TABLET;ORAL | 078447-001 | Aug 12, 2011 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Guardian Drug | LORATADINE | loratadine | TABLET;ORAL | 207569-001 | Mar 12, 2019 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


