Taro Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TARO, and what generic alternatives to TARO drugs are available?
TARO has two hundred and fifty-three approved drugs.
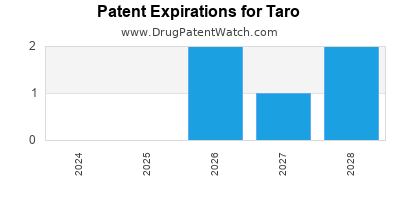
There are eight US patents protecting TARO drugs. There are four tentative approvals on TARO drugs.
There are thirty-one patent family members on TARO drugs in twelve countries and one hundred and seventy-three supplementary protection certificates in fourteen countries.
Summary for Taro
| International Patents: | 31 |
| US Patents: | 8 |
| Tradenames: | 133 |
| Ingredients: | 116 |
| NDAs: | 253 |
| Patent Litigation for Taro: | See patent lawsuits for Taro |
| PTAB Cases with Taro as petitioner: | See PTAB cases with Taro as petitioner |
Drugs and US Patents for Taro
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taro | CLOBETASOL PROPIONATE | clobetasol propionate | AEROSOL, FOAM;TOPICAL | 208779-001 | Oct 4, 2018 | AB1 | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Taro | MINOXIDIL (FOR WOMEN) | minoxidil | AEROSOL, FOAM;TOPICAL | 209074-002 | Apr 22, 2019 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Taro | FLUOCINONIDE | fluocinonide | CREAM;TOPICAL | 071500-001 | Jun 10, 1987 | AB1 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Taro | NORTRIPTYLINE HYDROCHLORIDE | nortriptyline hydrochloride | SOLUTION;ORAL | 077965-001 | Jun 20, 2006 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Taro | TOPICORT | desoximetasone | GEL;TOPICAL | 074904-001 | Jul 14, 1998 | AB | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Taro | DOXEPIN HYDROCHLORIDE | doxepin hydrochloride | CAPSULE;ORAL | 213063-003 | Jul 1, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Taro
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Taro | FLO-PRED | prednisolone acetate | SUSPENSION;ORAL | 022067-002 | Jan 17, 2008 | 6,656,482 | ⤷ Try a Trial |
| Taro | FLO-PRED | prednisolone acetate | SUSPENSION;ORAL | 022067-001 | Jan 17, 2008 | 5,881,926 | ⤷ Try a Trial |
| Taro | TOPICORT | desoximetasone | SPRAY;TOPICAL | 204141-001 | Apr 11, 2013 | 5,990,100 | ⤷ Try a Trial |
| Taro | PLIAGLIS | lidocaine; tetracaine | CREAM;TOPICAL | 021717-001 | Jun 29, 2006 | 5,919,479 | ⤷ Try a Trial |
| Taro | FLO-PRED | prednisolone acetate | SUSPENSION;ORAL | 022067-002 | Jan 17, 2008 | 6,071,523 | ⤷ Try a Trial |
| Taro | FLO-PRED | prednisolone acetate | SUSPENSION;ORAL | 022067-002 | Jan 17, 2008 | 5,881,926 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for TARO drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Topical Spray | 0.25% | ➤ Subscribe | 2013-12-18 |
| ➤ Subscribe | Topical Lotion | 0.5% | ➤ Subscribe | 2011-03-16 |
International Patents for Taro Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 103087095 | ⤷ Try a Trial |
| China | 101287374 | ⤷ Try a Trial |
| European Patent Office | 1909798 | ⤷ Try a Trial |
| Brazil | PI0612686 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2007019184 | ⤷ Try a Trial |
| Japan | 2009500422 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Taro Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0788511 | SPC/GB08/036 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MICAFUNGIN AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF IN PARTICULAR MICAFUNGIN SODIUM; REGISTERED: UK EU/1/08/448/001 20080425; UK EU/1/08/448/002 20080425 |
| 0031058 | 98C0008 | Belgium | ⤷ Try a Trial | PRODUCT NAME: TAZAROTENE; NAT. REGISTRATION NO/DATE: NL22604 19970922; FIRST REGISTRATION: DE - 37393.00.00 19961203 |
| 1304992 | PA2013025 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: CLINFAMYCINUM + TRETINOINUM; REGISTRATION NO/DATE: LT/1/13/3279/001, 2013 04 26 LT/1/13/3279/002, 2013 04 26 PA1332/043/001 20130323 |
| 2506844 | 1890025-8 | Sweden | ⤷ Try a Trial | PRODUCT NAME: COMBINATION OF A PHARMACEUTICALLY ACCEPTABLE SALT OF UMECLIDINIUM (E.G. UMECLIDINIUM BROMIDE), VILANTEROL OR A PHARMACEUTICALLY ACCEPTABLE SALT THEROF (E.G. VILANTEROL TRIFENATATE) AND FLUTICASONE FUROATE.; REG. NO/DATE: EU/1/17/1236 20171117 |
| 0473687 | 99C0008 | Belgium | ⤷ Try a Trial | PRODUCT NAME: FOSPHENYTOINUM DINATRICUM; REGISTRATION NO/DATE: 19 IS 102 F12 19980901; FIRST REGISTRATION: GB PL00019/1057 19980204 |
| 1948158 | 16C0018 | France | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN,SOUS FORME DE COMPLEXE SODIQUE SACUBITRIL VALSARTAN,C'EST-A-DIRE DE (3-((1S,3R)-1-BIPHENYL-4-YLMETHYL-3-ETHOXYCARBONYL-1-BUTYLCARBAMOYL) PROPIONATE-(S)-3'-METHYL-2'-(PENTANOY(2''-(TETRAZOL-5-YLATE)BIPHENYL-4'-YLMETHYL)AMINO)BUTYRATE)DE TRISODIUM HEMIPENTAHYDRATE; REGISTRATION NO/DATE: EU/1/15/1058 20151123 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.