VOQUEZNA DUAL PAK Drug Patent Profile
✉ Email this page to a colleague
When do Voquezna Dual Pak patents expire, and when can generic versions of Voquezna Dual Pak launch?
Voquezna Dual Pak is a drug marketed by Phathom and is included in one NDA. There are two patents protecting this drug.
This drug has one hundred and three patent family members in forty-three countries.
The generic ingredient in VOQUEZNA DUAL PAK is amoxicillin; vonoprazan fumarate. There are forty-six drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the amoxicillin; vonoprazan fumarate profile page.
DrugPatentWatch® Generic Entry Outlook for Voquezna Dual Pak
Voquezna Dual Pak will be eligible for patent challenges on May 3, 2026. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 3, 2032. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for VOQUEZNA DUAL PAK
| International Patents: | 103 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for VOQUEZNA DUAL PAK |
| What excipients (inactive ingredients) are in VOQUEZNA DUAL PAK? | VOQUEZNA DUAL PAK excipients list |
| DailyMed Link: | VOQUEZNA DUAL PAK at DailyMed |
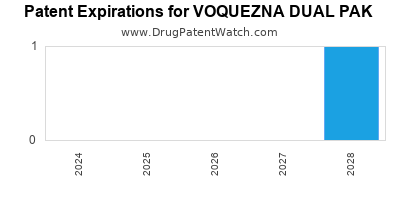

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VOQUEZNA DUAL PAK
Generic Entry Date for VOQUEZNA DUAL PAK*:
Constraining patent/regulatory exclusivity:
GENERATING ANTIBIOTIC INCENTIVES NOW NDA:
Dosage:
CAPSULE, TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Anatomical Therapeutic Chemical (ATC) Classes for VOQUEZNA DUAL PAK
US Patents and Regulatory Information for VOQUEZNA DUAL PAK
VOQUEZNA DUAL PAK is protected by two US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VOQUEZNA DUAL PAK is ⤷ Try a Trial.
This potential generic entry date is based on GENERATING ANTIBIOTIC INCENTIVES NOW.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting VOQUEZNA DUAL PAK
1-heterocyclylsulfonyl, 2-aminomethyl, 5-(hetero-) aryl substituted 1-H-pyrrole derivatives as acid secretion inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Pharmaceutical composition
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting VOQUEZNA DUAL PAK
NEW CHEMICAL ENTITY
Exclusivity Expiration: ⤷ Try a Trial
GENERATING ANTIBIOTIC INCENTIVES NOW
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phathom | VOQUEZNA DUAL PAK | amoxicillin; vonoprazan fumarate | CAPSULE, TABLET;ORAL | 215153-001 | May 3, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Phathom | VOQUEZNA DUAL PAK | amoxicillin; vonoprazan fumarate | CAPSULE, TABLET;ORAL | 215153-001 | May 3, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Phathom | VOQUEZNA DUAL PAK | amoxicillin; vonoprazan fumarate | CAPSULE, TABLET;ORAL | 215153-001 | May 3, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Phathom | VOQUEZNA DUAL PAK | amoxicillin; vonoprazan fumarate | CAPSULE, TABLET;ORAL | 215153-001 | May 3, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for VOQUEZNA DUAL PAK
When does loss-of-exclusivity occur for VOQUEZNA DUAL PAK?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2842
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 09277443
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0916689
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 32243
Estimated Expiration: ⤷ Try a Trial
Patent: 36400
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 11000170
Estimated Expiration: ⤷ Try a Trial
Patent: 13000224
Estimated Expiration: ⤷ Try a Trial
China
Patent: 2164581
Estimated Expiration: ⤷ Try a Trial
Patent: 2743330
Estimated Expiration: ⤷ Try a Trial
Patent: 4784180
Estimated Expiration: ⤷ Try a Trial
Colombia
Patent: 90638
Estimated Expiration: ⤷ Try a Trial
Costa Rica
Patent: 110110
Estimated Expiration: ⤷ Try a Trial
Dominican Republic
Patent: 011000033
Estimated Expiration: ⤷ Try a Trial
Patent: 013000172
Estimated Expiration: ⤷ Try a Trial
Ecuador
Patent: 11010855
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 1170272
Estimated Expiration: ⤷ Try a Trial
Patent: 1201451
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 09985
Estimated Expiration: ⤷ Try a Trial
Patent: 64833
Estimated Expiration: ⤷ Try a Trial
Georgia, Republic of
Patent: 0146122
Estimated Expiration: ⤷ Try a Trial
Patent: 0146166
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 95356
Estimated Expiration: ⤷ Try a Trial
Patent: 93363
Estimated Expiration: ⤷ Try a Trial
Patent: 11529445
Estimated Expiration: ⤷ Try a Trial
Patent: 13047239
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 6305
Estimated Expiration: ⤷ Try a Trial
Patent: 9461
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 11000757
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 556
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 1344
Estimated Expiration: ⤷ Try a Trial
Patent: 2592
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 110591
Estimated Expiration: ⤷ Try a Trial
Patent: 140977
Estimated Expiration: ⤷ Try a Trial
Patent: 141585
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 6685
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1101198
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 110042334
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 60962
Estimated Expiration: ⤷ Try a Trial
Patent: 69592
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 1010992
Estimated Expiration: ⤷ Try a Trial
Patent: 1431552
Estimated Expiration: ⤷ Try a Trial
Tunisia
Patent: 11000030
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 3332
Estimated Expiration: ⤷ Try a Trial
Uruguay
Patent: 008
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VOQUEZNA DUAL PAK around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Peru | 20070540 | DERIVADOS DE PIRROL COMO INHIBIDORES DE LA SECRECION ACIDA | ⤷ Try a Trial |
| Japan | 5295356 | ⤷ Try a Trial | |
| Serbia | 51697 | DERIVATI 1-HETEROCIKLILSULFONIL, 2-AMINOMETIL, 5-(HETERO-)ARIL SUPSTITUISANOG 1-H-PIROLA KAO INHIBITORI SEKRECIJE KISELINE (1-HETEROCYCLYLSULFONYL, 2-AMINOMETHYL, 5-(HETERO-)ARYL SUBSTITUTED 1-H-PYRROLE DERIVATIVES AS ACID SECRETION INHIBITORS) | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2010013823 | ⤷ Try a Trial | |
| South Africa | 200802488 | 1-heterocyclylsulfonyl, 2-aminomethyl, 5- (hetero-) aryl substituted 1-h-pyrrole derivatives as acid secretion inhibitors | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


