TEGSEDI Drug Patent Profile
✉ Email this page to a colleague
When do Tegsedi patents expire, and what generic alternatives are available?
Tegsedi is a drug marketed by Akcea Theraps and is included in one NDA. There are four patents protecting this drug.
This drug has thirty-four patent family members in twenty-four countries.
The generic ingredient in TEGSEDI is inotersen sodium. One supplier is listed for this compound. Additional details are available on the inotersen sodium profile page.
DrugPatentWatch® Generic Entry Outlook for Tegsedi
Tegsedi was eligible for patent challenges on October 5, 2022.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 29, 2031. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for TEGSEDI
| International Patents: | 34 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 2 |
| Drug Prices: | Drug price information for TEGSEDI |
| What excipients (inactive ingredients) are in TEGSEDI? | TEGSEDI excipients list |
| DailyMed Link: | TEGSEDI at DailyMed |
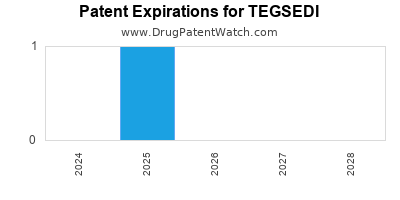

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TEGSEDI
Generic Entry Date for TEGSEDI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;SUBCUTANEOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TEGSEDI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Akcea Therapeutics | Phase 3 |
| Ionis Pharmaceuticals, Inc. | Phase 3 |
| GlaxoSmithKline | Phase 2/Phase 3 |
Pharmacology for TEGSEDI
| Drug Class | Antisense Oligonucleotide Transthyretin-directed RNA Interaction |
| Physiological Effect | Decreased RNA Integrity Increased Protein Breakdown |
Anatomical Therapeutic Chemical (ATC) Classes for TEGSEDI
US Patents and Regulatory Information for TEGSEDI
TEGSEDI is protected by four US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TEGSEDI is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting TEGSEDI
Modulation of transthyretin expression
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Diagnosis and treatment of disease
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Modulation of transthyretin expression
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Modulation of transthyretin expression
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF POLYNEUROPATHY OF HEREDITARY TRANSTHYRETIN AMYLOIDOSIS
FDA Regulatory Exclusivity protecting TEGSEDI
INDICATED FOR THE TREATMENT OF THE POLYNEUROPATHY OF HEREDITARY TRANSTHYRETIN-MEDIATED AMYLOIDOSIS IN ADULTS
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TEGSEDI
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | ⤷ Try a Trial | ⤷ Try a Trial |
| Akcea Theraps | TEGSEDI | inotersen sodium | SOLUTION;SUBCUTANEOUS | 211172-001 | Oct 5, 2018 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TEGSEDI
When does loss-of-exclusivity occur for TEGSEDI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Brazil
Patent: 2012027547
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 97792
Estimated Expiration: ⤷ Try a Trial
Patent: 94063
Estimated Expiration: ⤷ Try a Trial
China
Patent: 3038345
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0170737
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 19070
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 31909
Estimated Expiration: ⤷ Try a Trial
Patent: 900001
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 2697
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 96175
Estimated Expiration: ⤷ Try a Trial
Patent: 13526860
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 563920
Estimated Expiration: ⤷ Try a Trial
Patent: 2019001
Estimated Expiration: ⤷ Try a Trial
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
Luxembourg
Patent: 0096
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 3559
Estimated Expiration: ⤷ Try a Trial
Patent: 12012624
Estimated Expiration: ⤷ Try a Trial
Netherlands
Patent: 0963
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 3339
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 19001
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 92669
Estimated Expiration: ⤷ Try a Trial
Patent: 12150394
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 011
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 63920
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1835386
Estimated Expiration: ⤷ Try a Trial
Patent: 130098162
Estimated Expiration: ⤷ Try a Trial
Patent: 180026798
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 25689
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TEGSEDI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Australia | 2003236510 | ⤷ Try a Trial | |
| European Patent Office | 0831854 | ⤷ Try a Trial | |
| Australia | 1998692 | ⤷ Try a Trial | |
| European Patent Office | 1163373 | ⤷ Try a Trial | |
| Australia | 4684099 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TEGSEDI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2563920 | 122019000001 | Germany | ⤷ Try a Trial | PRODUCT NAME: VERBINDUNG, UMFASSEND EIN MODIFIZIERTES OLIGONUKLEOTID MIT EINER NUKLEOBASENSEQUENZ BESTEHEND AUS 20 VERKNUEPFTEN NUKLEOSIDEN GEMAESS EP B1 2563920, ANSPRUCH 1 (SEQ ID NO: 80), WOBEI DAS MODIFIZIERTE OLIGONUKLEOTID UMFASST: EIN LUECKENSEGMENT, DAS AUS ZEHN VERKNUEPFTEN DESOXYNUKLEOSIDEN BESTEHT ; EIN 5'-FLUEGELSEGMENT BESTEHEND AUS FUENF VERKNUEPFTEN NUKLEOSIDEN; UND EIN 3'-FLUEGELSEGMENT BESTEHEND AUS FUEN; REGISTRATION NO/DATE: EU/1/18/1296 20180706 |
| 2563920 | C201930001 | Spain | ⤷ Try a Trial | PRODUCT NAME: INOTERSEN Y SUS SALES FARMACEUTICAMENTE ACEPTABLES, EN PARTICULAR LA SAL SODICA; NATIONAL AUTHORISATION NUMBER: EU/1/18/1296; DATE OF AUTHORISATION: 20180706; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/18/1296; DATE OF FIRST AUTHORISATION IN EEA: 20180706 |
| 2563920 | 2019001 | Norway | ⤷ Try a Trial | PRODUCT NAME: FORBINDELSE OMFATTENDE ET MODIFISERT OLIGONUKLEOTID SOM HAR EN NUKLEOBASESEKVENS BESTAENDE AV 20 KOBLEDE NUKLEOSIDER IFOELGE EP B1 2563920 KRAV 1 (SEQ ID NO: 80), HVOR DET MODIFISERTE OLIGONUKLEOTIDET OMFATTER: ET AVSTANDSSEGMENT BESTAENDE AV TI KOBLEDE DEOKSYNUKLEOSIDER; ET 5'- VINGESEGMENT BESTAENDE AV FEM KOBLEDE NUKLEOSIDER; OG ET 3'-VINGESEGMENT BESTAENDE AV FEM KOBLEDE NUKLEOSIDER; HVOR AVSTANDSSEGMENTET ER PLASSERT MELLOM 5'- VINGESEGMENTET OG 3'- VINGESEGMENTET, HVOR HVERT NUKLEOSID AV HVERT VINGESEGMENT OMFATTER ET 2'- 0-METOKSYETYLSUKKER; HVOR HVER CYTOSIN AV DET MODIFISERTE OLIGONUKLEOTID ER EN 5-METYLCYTOSIN, OG HVOR HVER INTERNUKLEOSID-BINDING AV DET MODIFISERTE OL |
| 2563920 | 354 1-2019 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: INOTERSEN VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/18/1296 20180710 |
| 2563920 | CA 2019 00001 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMPOUND COMPRISING A MODIFIED OLIGONUCLEOTIDE HAVING A NUCLEOBASE SEQUENCE CONSISTING OF 20 LINKED NUCLEOSIDES ACCORDING TO EP B1 2563920 CLAIM 1 (SEQ ID NO: 80), ...SPECIFICALLY INOTERSEN; AND ITS DERIVATIVES, INCLUDING SODIUM SALTS ...; REG. NO/DATE: EU/1/18/1296 20180710 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


