KOSELUGO Drug Patent Profile
✉ Email this page to a colleague
When do Koselugo patents expire, and when can generic versions of Koselugo launch?
Koselugo is a drug marketed by Astrazeneca and is included in one NDA. There are five patents protecting this drug.
This drug has one hundred and ninety-eight patent family members in forty-four countries.
The generic ingredient in KOSELUGO is selumetinib sulfate. One supplier is listed for this compound. Additional details are available on the selumetinib sulfate profile page.
DrugPatentWatch® Generic Entry Outlook for Koselugo
Koselugo was eligible for patent challenges on April 10, 2024.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 10, 2027. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for KOSELUGO
| International Patents: | 198 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 129 |
| Clinical Trials: | 11 |
| Patent Applications: | 2,416 |
| Drug Prices: | Drug price information for KOSELUGO |
| What excipients (inactive ingredients) are in KOSELUGO? | KOSELUGO excipients list |
| DailyMed Link: | KOSELUGO at DailyMed |
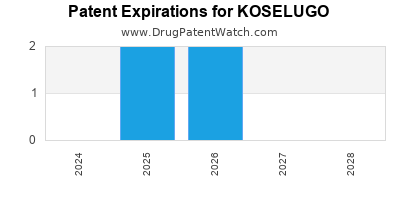

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for KOSELUGO
Generic Entry Date for KOSELUGO*:
Constraining patent/regulatory exclusivity:
INDICATED FOR THE TREATMENT OF PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER WITH NEUROFIBROMATOSIS TYPE 1 (NF1) WHO HAVE SYMPTOMATIC, INOPERABLE PLEXIFORM NEUROFIBROMAS (PN) NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for KOSELUGO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| AstraZeneca | Phase 1 |
| Merck Sharp & Dohme Corp. | Phase 1 |
| National Cancer Institute (NCI) | Phase 3 |
Pharmacology for KOSELUGO
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Mitogen-Activated Protein Kinase Kinase 1 Inhibitors Mitogen-Activated Protein Kinase Kinase 2 Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for KOSELUGO
US Patents and Regulatory Information for KOSELUGO
KOSELUGO is protected by five US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of KOSELUGO is ⤷ Try a Trial.
This potential generic entry date is based on INDICATED FOR THE TREATMENT OF PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER WITH NEUROFIBROMATOSIS TYPE 1 (NF1) WHO HAVE SYMPTOMATIC, INOPERABLE PLEXIFORM NEUROFIBROMAS (PN).
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting KOSELUGO
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
N3 alkylated benzimidazole derivatives as MEK inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
N3 alkylated benzimidazole derivatives as MEK inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Hydrogen sulfate salt
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Hydrogen sulfate salt
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER WITH NEUROFIBROMATOSIS TYPE 1 (NF1) WHO HAVE SYMPTOMATIC, INOPERABLE PLEXIFORM NEUROFIBROMAS (PN)
FDA Regulatory Exclusivity protecting KOSELUGO
NEW CHEMICAL ENTITY
Exclusivity Expiration: ⤷ Try a Trial
INDICATED FOR THE TREATMENT OF PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER WITH NEUROFIBROMATOSIS TYPE 1 (NF1) WHO HAVE SYMPTOMATIC, INOPERABLE PLEXIFORM NEUROFIBROMAS (PN)
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-001 | Apr 10, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-001 | Apr 10, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-001 | Apr 10, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-002 | Apr 10, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for KOSELUGO
When does loss-of-exclusivity occur for KOSELUGO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 8696
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 06330759
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0620091
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 34149
Estimated Expiration: ⤷ Try a Trial
China
Patent: 1360718
Estimated Expiration: ⤷ Try a Trial
Patent: 2329270
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0130663
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 14303
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 68948
Estimated Expiration: ⤷ Try a Trial
Ecuador
Patent: 088597
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 68948
Estimated Expiration: ⤷ Try a Trial
France
Patent: C1051
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 24043
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 100046
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 2224
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 27723
Estimated Expiration: ⤷ Try a Trial
Patent: 09521487
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 968948
Estimated Expiration: ⤷ Try a Trial
Patent: 2021530
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 7733
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 08008298
Estimated Expiration: ⤷ Try a Trial
Netherlands
Patent: 1139
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 9792
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 68948
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 68948
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 18790
Estimated Expiration: ⤷ Try a Trial
Patent: 08129199
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 843
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 68948
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 0805705
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1361460
Estimated Expiration: ⤷ Try a Trial
Patent: 080080200
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 21746
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 05756
Estimated Expiration: ⤷ Try a Trial
Patent: 0800915
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 531
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering KOSELUGO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Argentina | 038971 | DERIVADOS DE BENCIMIDAZOL N3 ALQUILADO COMO INHIBIDORES DE MEK | ⤷ Try a Trial |
| Hungary | S2100046 | ⤷ Try a Trial | |
| Croatia | P20120657 | ⤷ Try a Trial | |
| Dominican Republic | P2013000023 | UN COMPUESTO SELECCIONADO DEL ACIDO 6-(4-BROMO-2-CLORO-FENILAMINO) -7-FLUROR-3-METIL-3H-BENZOIMIDAZOL -5-CARBOXILICO (2-HIDROXI-ETOXI)-AMIDA Y SALES FARMACEUTICAMENTE ACEPTABLES DEL MISMO, Y COMPOSICIONES FARMACEUTICAS QUE CONTIENEN DICHO COMPUESTO | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for KOSELUGO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1968948 | CA 2021 00044 | Denmark | ⤷ Try a Trial | PRODUCT NAME: SELUMETINIB, INKLUSIV FARMACEUTISK ACCEPTABLE SALTE (ISAER HYDROGENSULFAT), ESTRE, SOLVATER OG ENANTIOMERE DERAF; REG. NO/DATE: EU/1/21/1552 20210619 |
| 1482932 | 648 | Finland | ⤷ Try a Trial | |
| 1482932 | PA2019007,C1482932 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: BINIMETINIBAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA ARBA SOLVATAS; REGISTRATION NO/DATE: EU/1/18/1315 20180920 |
| 1482932 | CA 2019 00011 | Denmark | ⤷ Try a Trial | PRODUCT NAME: BINIMETINIB I ALLE FORMER BESKYTTET AF GRUNDPATENTET; REG. NO/DATE: EU/1/18/1315 20180924 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


