Salix Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SALIX, and what generic alternatives to SALIX drugs are available?
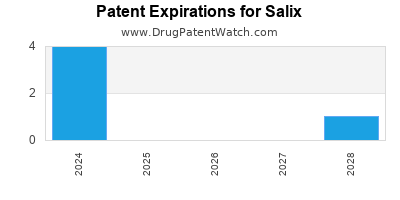
SALIX has eighteen approved drugs.
There are seventy-five US patents protecting SALIX drugs.
There are six hundred and forty-nine patent family members on SALIX drugs in fifty-five countries and ninety-seven supplementary protection certificates in twelve countries.
Summary for Salix
| International Patents: | 649 |
| US Patents: | 75 |
| Tradenames: | 15 |
| Ingredients: | 13 |
| NDAs: | 18 |
| Patent Litigation for Salix: | See patent lawsuits for Salix |
| PTAB Cases with Salix as patent owner: | See PTAB cases with Salix as patent owner |
Drugs and US Patents for Salix
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Salix | RELISTOR | methylnaltrexone bromide | TABLET;ORAL | 208271-001 | Jul 19, 2016 | RX | Yes | Yes | 9,724,343 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | DIURIL | chlorothiazide | SUSPENSION;ORAL | 011870-001 | Approved Prior to Jan 1, 1982 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Salix | FENOGLIDE | fenofibrate | TABLET;ORAL | 022118-001 | Aug 10, 2007 | AB | RX | Yes | No | 9,173,847 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Salix | TRULANCE | plecanatide | TABLET;ORAL | 208745-001 | Jan 19, 2017 | RX | Yes | Yes | 11,834,521 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-001 | May 25, 2004 | RX | Yes | Yes | 8,518,949 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Salix
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 8,552,025 | ⤷ Try a Trial |
| Salix | ZEGERID | omeprazole; sodium bicarbonate | FOR SUSPENSION;ORAL | 021636-002 | Dec 21, 2004 | 6,645,988 | ⤷ Try a Trial |
| Salix | UCERIS | budesonide | TABLET, EXTENDED RELEASE;ORAL | 203634-001 | Jan 14, 2013 | 8,784,888 | ⤷ Try a Trial |
| Salix | APRISO | mesalamine | CAPSULE, EXTENDED RELEASE;ORAL | 022301-001 | Oct 31, 2008 | 8,956,647 | ⤷ Try a Trial |
| Salix | UCERIS | budesonide | TABLET, EXTENDED RELEASE;ORAL | 203634-001 | Jan 14, 2013 | RE43799 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for SALIX drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsules | 0.375 g | ➤ Subscribe | 2012-04-03 |
| ➤ Subscribe | Powder for Oral Suspension | 40 mg/1680 mg per packet | ➤ Subscribe | 2007-08-24 |
| ➤ Subscribe | For Oral Solution | 140 g, 5.2 g, 2.2.g, 48.11 g, 9g and 7.54 g per pouch | ➤ Subscribe | 2018-12-06 |
| ➤ Subscribe | Injection | 12 mg/0.6 mL | ➤ Subscribe | 2015-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2016-09-06 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2010-08-24 |
| ➤ Subscribe | Tablets | 550 mg | ➤ Subscribe | 2015-12-18 |
| ➤ Subscribe | Extended-release Tablets | 9 mg | ➤ Subscribe | 2013-03-11 |
| ➤ Subscribe | Capsules | 20 mg/1100 mg and 40 mg/1100 mg | ➤ Subscribe | 2007-04-30 |
| ➤ Subscribe | Powder for Oral Suspension | 20mg/1680mg per packet | ➤ Subscribe | 2007-11-13 |
| ➤ Subscribe | Tablets | 40 mg and 120 mg | ➤ Subscribe | 2010-03-17 |
| ➤ Subscribe | Injection | 8 mg/0.4 mL | ➤ Subscribe | 2015-09-08 |
| ➤ Subscribe | For Oral Solution | 100 g, 7.5 g, 2.691 g, 1.015 g, 5.9 g and 4.7 g per pouch | ➤ Subscribe | 2007-11-27 |
| ➤ Subscribe | Tablets | 1.102 g and 0.398 g | ➤ Subscribe | 2008-04-09 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2019-01-28 |
International Patents for Salix Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Brazil | 112015005270 | ⤷ Try a Trial |
| Japan | 2015527385 | ⤷ Try a Trial |
| Denmark | 3141251 | ⤷ Try a Trial |
| Japan | 2013216710 | ⤷ Try a Trial |
| Spain | 2727728 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Salix Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1499331 | 13C0055 | France | ⤷ Try a Trial | PRODUCT NAME: SULFATE DE SODIUM ANHYDRE, SULFATE DE MAGNESIUM HEPTAHYDRATE, SULFATE DE POTASSIUM; NAT. REGISTRATION NO/DATE: NL41696 20130426; FIRST REGISTRATION: BE - 434323 20130220 |
| 2435024 | LUC00208 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE FORMOTEROL (Y COMPRIS SES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES), DE GLYCOPYRRONIUM (Y COMPRIS SES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES) ET DE BUDESONIDE (Y COMPRIS SES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES); AUTHORISATION NUMBER AND DATE: EU/1/20/1468 20201210 |
| 0809498 | SPC/GB10/012 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF ACYCLOVIR AND HYDROCORTISONE; REGISTERED: UK PL18191/0001-0001 20091112 |
| 1912999 | 2014/058 | Ireland | ⤷ Try a Trial | PRODUCT NAME: SIMEPREVIR, OR A SALT THEREOF, INCLUDING SIMEPREVIR SODIUM; REGISTRATION NO/DATE: EU/1/14/924/001-002 20140516 |
| 2822954 | SPC/GB18/031 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BICTEGRAVIR SODIUM.; REGISTERED: UK EU/1/18/1289/001(NI) 20180625; UK EU/1/18/1289/002(NI) 20180625; UK PLGB 11972/0008 20180625 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.