Par Pharm Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PAR PHARM INC, and what generic alternatives to PAR PHARM INC drugs are available?
PAR PHARM INC has thirty-eight approved drugs.
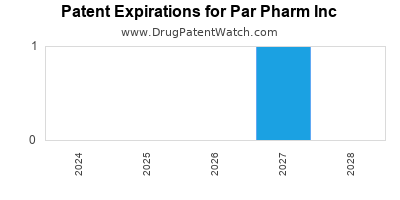
There is one US patent protecting PAR PHARM INC drugs. There are seven tentative approvals on PAR PHARM INC drugs.
There are nine patent family members on PAR PHARM INC drugs in eight countries and sixty-one supplementary protection certificates in fourteen countries.
Drugs and US Patents for Par Pharm Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Par Pharm Inc | OLMESARTAN MEDOXOMIL, AMLODIPINE AND HYDROCHLOROTHIAZIDE | amlodipine besylate; hydrochlorothiazide; olmesartan medoxomil | TABLET;ORAL | 206137-002 | Oct 26, 2016 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | ASPIRIN AND DIPYRIDAMOLE | aspirin; dipyridamole | CAPSULE, EXTENDED RELEASE;ORAL | 207944-001 | Jan 18, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | MIDODRINE HYDROCHLORIDE | midodrine hydrochloride | TABLET;ORAL | 207169-001 | Oct 29, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | DEXMETHYLPHENIDATE HYDROCHLORIDE | dexmethylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 202842-001 | Nov 30, 2016 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | COLCHICINE | colchicine | CAPSULE;ORAL | 208678-001 | Nov 29, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Par Pharm Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 101163679 | ⤷ Try a Trial |
| European Patent Office | 1948613 | ⤷ Try a Trial |
| Australia | 2006240490 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2006115626 | ⤷ Try a Trial |
| Japan | 2008538560 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Par Pharm Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2435025 | LUC00124 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: UNE COMBINAISON DE GLYCOPYRROLATE (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES) ET DE FORMOTEROL (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES); AUTHORISATION NUMBER AND DATE: EU/1/18/1339 20181220 |
| 0237929 | 68/1997 | Austria | ⤷ Try a Trial | PRODUCT NAME: TOLCAPONE; REGISTRATION NO/DATE: EU/1/97/044/001 - EU/1/97/044/006 19970827 |
| 2101777 | 2016C/032 | Belgium | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN EN COMBINAISON AVEC LE TADALAFIL; AUTHORISATION NUMBER AND DATE: EU/1/08/451 20151125 |
| 0443983 | C300445 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN, AMLODIPINE EN HYDROCHLOORTHIAZIDE EN FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN; REGISTRATION NO/DATE: EU/1/09/569/001-060 20091016 |
| 0450097 | 09C0049 | France | ⤷ Try a Trial | PRODUCT NAME: TOLVAPTAN, EVENTUELLEMENT SOUS FORME DE SEL; REGISTRATION NO/DATE: EU/1/09/539/001 20090803 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.