Ferring Pharms Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for FERRING PHARMS INC, and what generic alternatives to FERRING PHARMS INC drugs are available?
FERRING PHARMS INC has nine approved drugs.
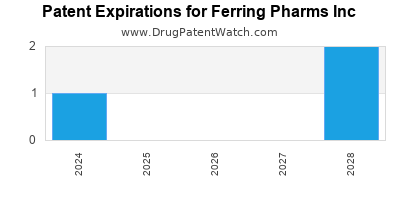
There are twelve US patents protecting FERRING PHARMS INC drugs.
There are one hundred and eighty-four patent family members on FERRING PHARMS INC drugs in thirty-nine countries and fourteen supplementary protection certificates in nine countries.
Summary for Ferring Pharms Inc
| International Patents: | 184 |
| US Patents: | 12 |
| Tradenames: | 9 |
| Ingredients: | 4 |
| NDAs: | 9 |
Drugs and US Patents for Ferring Pharms Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ferring Pharms Inc | STIMATE | desmopressin acetate | SPRAY, METERED;NASAL | 020355-001 | Mar 7, 1994 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ferring Pharms Inc | MILPROSA | progesterone | SYSTEM;VAGINAL | 201110-001 | Apr 29, 2020 | DISCN | Yes | No | 8,580,293 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ferring Pharms Inc | DDAVP | desmopressin acetate | SOLUTION;NASAL | 017922-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ferring Pharms Inc | DDAVP | desmopressin acetate | INJECTABLE;INJECTION | 018938-002 | Apr 25, 1995 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Ferring Pharms Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ferring Pharms Inc | DDAVP | desmopressin acetate | SPRAY, METERED;NASAL | 017922-002 | Feb 6, 1989 | 5,674,850 | ⤷ Try a Trial |
| Ferring Pharms Inc | DDAVP | desmopressin acetate | INJECTABLE;INJECTION | 018938-002 | Apr 25, 1995 | 5,763,407 | ⤷ Try a Trial |
| Ferring Pharms Inc | DDAVP | desmopressin acetate | TABLET;ORAL | 019955-002 | Sep 6, 1995 | 7,022,340 | ⤷ Try a Trial |
| Ferring Pharms Inc | DDAVP | desmopressin acetate | TABLET;ORAL | 019955-002 | Sep 6, 1995 | 5,763,407 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for FERRING PHARMS INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Solution | 10 mg, 3.5 g, and12 g | ➤ Subscribe | 2019-02-11 |
| ➤ Subscribe | Oral Solution | 10 mg, 3.5 g, and 12 g | ➤ Subscribe | 2014-05-21 |
International Patents for Ferring Pharms Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 6001121 | ⤷ Try a Trial |
| Lithuania | 3225250 | ⤷ Try a Trial |
| Norway | 20045345 | ⤷ Try a Trial |
| South Korea | 20130105757 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Ferring Pharms Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2782584 | LUC00245 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17SS-ESTRADIOL), EVENTUELLEMENT SOUS FORME D'UN SEL, HYDRATE OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI (Y COMPRIS SOUS FORME HEMIHYDRATEE), ET DE LA PROGESTERONE; AUTHORISATION NUMBER AND DATE: BE582231 20210701 |
| 3225249 | CA 2019 00023 | Denmark | ⤷ Try a Trial | PRODUCT NAME: DESMOPRESSIN ELLER ET ACETATSALT DERAF; NAT. REG. NO/DATE: 55858, 55859 (DK) 20160526; FIRST REG. NO/DATE: BE BE497271, BE497280 20160504 |
| 2712622 | LUC00015 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: DESMOPRESSINE OU UN DE SES SELS D'ACETATE; AUTHORISATION NUMBER AND DATE: 497271; 497280 20161101 |
| 2782584 | 2021C/558 | Belgium | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17--ESTRADIOL), EVENTUELLEMENT SOUS FORME D'UN SEL, HYDRATE OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI (Y COMPRIS SOUS FORME HEMIHYDRATEE), ET DE LA PROGESTERONE; AUTHORISATION NUMBER AND DATE: BE582231 20210406 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.