Exelixis Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for EXELIXIS INC, and when can generic versions of EXELIXIS INC drugs launch?
EXELIXIS INC has one approved drug.
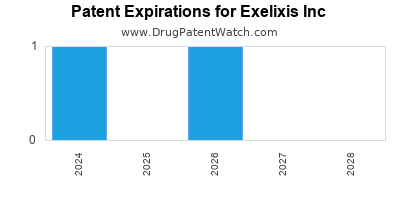
There are ten US patents protecting EXELIXIS INC drugs.
There are two hundred and seven patent family members on EXELIXIS INC drugs in thirty-two countries and nineteen supplementary protection certificates in sixteen countries.
Drugs and US Patents for Exelixis Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-002 | Apr 25, 2016 | RX | Yes | No | 11,298,349 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-003 | Apr 25, 2016 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-001 | Apr 25, 2016 | RX | Yes | No | 9,724,342 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-003 | Apr 25, 2016 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-003 | Apr 25, 2016 | RX | Yes | Yes | 9,724,342 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-001 | Apr 25, 2016 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Exelixis Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2016188215 | ⤷ Try a Trial |
| Australia | 2013203780 | ⤷ Try a Trial |
| Spain | 2402524 | ⤷ Try a Trial |
| South Korea | 101862324 | ⤷ Try a Trial |
| New Zealand | 614130 | ⤷ Try a Trial |
| New Zealand | 607118 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Exelixis Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2213661 | 300678 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/13/890/001-003 20140326 |
| 2213661 | 2014/052 | Ireland | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF.; REGISTRATION NO/DATE: EU/1/13/890/001-003 20140326 |
| 2213661 | 583 | Finland | ⤷ Try a Trial | |
| 2213661 | C02213661/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66471 15.12.2017 |
| 2213661 | 92508 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB ET TOUTES LES FORMES THERAPEUTIQUEMENT EQUIVALENTES QUI EN DERIVENT TELLES QUE PROTEGEES PAR LE BREVET DE BASE, Y COMPRIS LES SELS PHARMACEUTIQUEMENT ACCEPTABLES. FIRST REGISTRATION: 20140326 |
| 2213661 | C 2014 036 | Romania | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB SI ORICE FORMA ECHIVALENTA TERAPEUTIC AACESTUIA, INCLUSIV SARURILE ACCEPT DATE OF NATIONAL AUTHORISATION: 20140321; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/13/890/001, EU/1/13/890/002, EU/1/13/890/003; DATE OF FIRST AUTHORISATION IN EEA: 20140321 ABILE FARMACEUTIC; NATIONAL AUTHORISATION NUMBER: EU/1/13/890/001, EU/1/13/890/002, EU/1/13/890/003; |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.