Esperion Theraps Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ESPERION THERAPS INC, and what generic alternatives to ESPERION THERAPS INC drugs are available?
ESPERION THERAPS INC has two approved drugs.
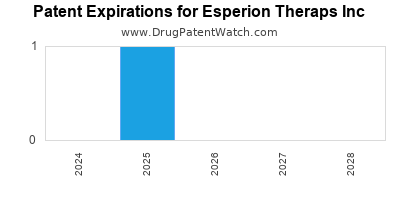
There are five US patents protecting ESPERION THERAPS INC drugs.
There are sixty-seven patent family members on ESPERION THERAPS INC drugs in twenty-three countries and seventeen supplementary protection certificates in sixteen countries.
Summary for Esperion Theraps Inc
| International Patents: | 67 |
| US Patents: | 5 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for Esperion Theraps Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Esperion Theraps Inc | NEXLIZET | bempedoic acid; ezetimibe | TABLET;ORAL | 211617-001 | Feb 26, 2020 | RX | Yes | Yes | 10,912,751 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Esperion Theraps Inc | NEXLIZET | bempedoic acid; ezetimibe | TABLET;ORAL | 211617-001 | Feb 26, 2020 | RX | Yes | Yes | 7,335,799 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Esperion Theraps Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | 9,624,152 | ⤷ Try a Trial |
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | 8,497,301 | ⤷ Try a Trial |
| Esperion Theraps Inc | NEXLIZET | bempedoic acid; ezetimibe | TABLET;ORAL | 211617-001 | Feb 26, 2020 | 8,497,301 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Esperion Theraps Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2020257571 | ⤷ Try a Trial |
| Japan | 2021193105 | ⤷ Try a Trial |
| Australia | 2003299993 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Esperion Theraps Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2404890 | 20C1041 | France | ⤷ Try a Trial | PRODUCT NAME: ACIDE BEMPEDOIQUE, OU UN SEL, HYDRATE, PRODUIT DE SOLVATATION OU MELANGE DE CEUX-CI, PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE: EU/1/20/1424 20200331 |
| 2404890 | C 2020 031 | Romania | ⤷ Try a Trial | PRODUCT NAME: ACID BEMPEDOIC, SAU O SARE, HIDRAT, SOLVAT ACCEPTABILE FARMACEUTIC SAU AMESTECUL ACESTORA; NATIONAL AUTHORISATION NUMBER: EU/1/20/1424; DATE OF NATIONAL AUTHORISATION: 20200327; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1424; DATE OF FIRST AUTHORISATION IN EEA: 20200327 |
| 2404890 | CA 2020 00041 | Denmark | ⤷ Try a Trial | PRODUCT NAME: BEMPEDOINSYRE ELLER ET/EN FARMACEUTISK ACCEPTABEL(T) SALT, HYDRAT, SOLVAT ELLER BLANDING DERAF; REG. NO/DATE: EU/1/20/1424 20200331 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.