Chemo Research Sl Company Profile
✉ Email this page to a colleague
What is the competitive landscape for CHEMO RESEARCH SL, and what generic alternatives to CHEMO RESEARCH SL drugs are available?
CHEMO RESEARCH SL has three approved drugs.
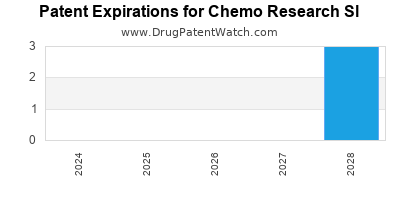
There are seven US patents protecting CHEMO RESEARCH SL drugs.
There are sixteen patent family members on CHEMO RESEARCH SL drugs in nine countries and forty-one supplementary protection certificates in fourteen countries.
Summary for Chemo Research Sl
| International Patents: | 16 |
| US Patents: | 7 |
| Tradenames: | 3 |
| Ingredients: | 3 |
| NDAs: | 3 |
Drugs and US Patents for Chemo Research Sl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemo Research Sl | NUVESSA | metronidazole | GEL;VAGINAL | 205223-001 | Mar 24, 2014 | AB | RX | Yes | Yes | 8,658,678 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Chemo Research Sl | NUVESSA | metronidazole | GEL;VAGINAL | 205223-001 | Mar 24, 2014 | AB | RX | Yes | Yes | 8,946,276 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Chemo Research Sl | NUVESSA | metronidazole | GEL;VAGINAL | 205223-001 | Mar 24, 2014 | AB | RX | Yes | Yes | 7,893,097 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Chemo Research Sl | ESTRADIOL | estradiol | GEL;TRANSDERMAL | 211783-001 | Aug 10, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Chemo Research Sl | BENZNIDAZOLE | benznidazole | TABLET;ORAL | 209570-002 | Aug 29, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Chemo Research Sl | NUVESSA | metronidazole | GEL;VAGINAL | 205223-001 | Mar 24, 2014 | AB | RX | Yes | Yes | 8,877,792 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Chemo Research Sl Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2016200825 | ⤷ Try a Trial |
| Japan | 6235087 | ⤷ Try a Trial |
| Brazil | 112013033759 | ⤷ Try a Trial |
| Mexico | 2014000066 | ⤷ Try a Trial |
| Australia | 2012275292 | ⤷ Try a Trial |
| China | 107823123 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Chemo Research Sl Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0136011 | 99C0003 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL AND NORETHISTERONE; FIRST REGISTRATION NO/DATE: 403 IS 106 F3 19980928; FIRST REGISTRATION: SE 14007 19980306 |
| 0584952 | 99C0004 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL, HEMIHYDRATE, NORETHISTERONE, ACETATE; NAT. REGISTRATION NO/DATE: NL 23753 19981210; FIRST REGISTRATION: SE - 14 007 19980306 |
| 0770388 | 2009/012 | Ireland | ⤷ Try a Trial | PRODUCT NAME: QLAIRA-ESTRADIOL VALERATE/DIENOGEST; NAT REGISTRATION NO/DATE: PA1410/58/1 20090109; FIRST REGISTRATION NO/DATE: BE327792 20081103 |
| 0398460 | SPC/GB04/032 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL, OPTIONALLY IN THE FORM OF A HYDRATE, TOGETHER WITH DROSPIRENONE; REGISTERED: NL RVG 27505 20021211; UK PL 00053/0341 20040310 |
| 0770388 | 09C0018 | France | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL VALERATE; DIENOGEST; NAT. REGISTRATION NO/DATE: NL35170 20081210; FIRST REGISTRATION: BE327792 20081103 |
| 2782584 | LUC00245 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17SS-ESTRADIOL), EVENTUELLEMENT SOUS FORME D'UN SEL, HYDRATE OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI (Y COMPRIS SOUS FORME HEMIHYDRATEE), ET DE LA PROGESTERONE; AUTHORISATION NUMBER AND DATE: BE582231 20210701 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.