Cardinal Health 414 Company Profile
✉ Email this page to a colleague
What is the competitive landscape for CARDINAL HEALTH 414, and what generic alternatives to CARDINAL HEALTH 414 drugs are available?
CARDINAL HEALTH 414 has five approved drugs.
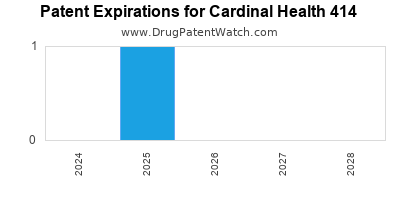
There are two US patents protecting CARDINAL HEALTH 414 drugs.
There are thirty patent family members on CARDINAL HEALTH 414 drugs in twelve countries and seventy-one supplementary protection certificates in nine countries.
Summary for Cardinal Health 414
| International Patents: | 30 |
| US Patents: | 2 |
| Tradenames: | 5 |
| Ingredients: | 5 |
| NDAs: | 5 |
Drugs and US Patents for Cardinal Health 414
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cardinal Health 414 | TECHNETIUM TC-99M MEDRONATE KIT | technetium tc-99m medronate kit | INJECTABLE;INJECTION | 018107-001 | Approved Prior to Jan 1, 1982 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Cardinal Health 414 | LYMPHOSEEK KIT | technetium tc-99m tilmanocept | INJECTABLE;INJECTION | 202207-001 | Mar 13, 2013 | RX | Yes | Yes | 6,409,990 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Cardinal Health 414 | SODIUM FLUORIDE F-18 | sodium fluoride f-18 | INJECTABLE;INTRAVENOUS | 203780-001 | Jul 30, 2015 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Cardinal Health 414 | LYMPHOSEEK KIT | technetium tc-99m tilmanocept | INJECTABLE;INJECTION | 202207-001 | Mar 13, 2013 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Cardinal Health 414 Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2015164933 | ⤷ Try a Trial |
| European Patent Office | 3884965 | ⤷ Try a Trial |
| China | 102301429 | ⤷ Try a Trial |
| Australia | 2010208624 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Cardinal Health 414 Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2822954 | 1890030-8 | Sweden | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BICTEGRAVIR SODIUM; REG. NO/DATE: EU/1/18/1289 20180625 |
| 2666774 | CR 2020 00037 | Denmark | ⤷ Try a Trial | PRODUCT NAME: RELEBACTAM, OPTIONALLY IN THE FORM OF THE MONOHYDRATE, IMIPENEM AND CILASTATIN, OPTIONALLY IN THE FORM OF THE SODIUM SALT; REG. NO/DATE: EU/1/19/1420 20200217 |
| 0806968 | SPC/GB07/011 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: GADOFOSVESET TRISODIUM; REGISTERED: UK EU/1/05/313/001 20051003; UK EU/1/05/313/002 20051003; UK EU/1/05/313/003 20051003; UK EU/1/05/313/004 20051003; UK EU/1/05/313/005 20051003; UK EU/1/05/313/006 20051003; UK EU/1/05/313/007 20051003; UK EU/1/05/313/008 20051003; UK EU/1/05/313/009 20051003 |
| 2203431 | 1590018-6 | Sweden | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REG. NO/DATE: EU/1/14/983 20150119 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.