CIPLA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for CIPLA, and when can generic versions of CIPLA drugs launch?
CIPLA has ninety-two approved drugs.
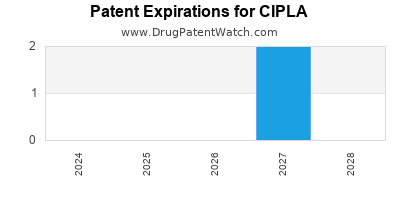
There are six US patents protecting CIPLA drugs. There are thirty-three tentative approvals on CIPLA drugs.
There are thirty patent family members on CIPLA drugs in twenty-one countries and four hundred and thirty-nine supplementary protection certificates in seventeen countries.
Summary for CIPLA
| International Patents: | 30 |
| US Patents: | 6 |
| Tradenames: | 81 |
| Ingredients: | 81 |
| NDAs: | 92 |
| Patent Litigation for CIPLA: | See patent lawsuits for CIPLA |
| PTAB Cases with CIPLA as petitioner: | See PTAB cases with CIPLA as petitioner |
Drugs and US Patents for CIPLA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cipla | SODIUM NITROPRUSSIDE | sodium nitroprusside | INJECTABLE;INJECTION | 210855-001 | Jul 16, 2018 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Cipla | TADALAFIL | tadalafil | TABLET;ORAL | 209539-003 | Mar 26, 2019 | AB1 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Cipla | DIMETHYL FUMARATE | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 210305-002 | Sep 24, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for CIPLA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Slovenia | 2217610 | ⤷ Try a Trial |
| China | 101516370 | ⤷ Try a Trial |
| China | 101868472 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for CIPLA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1874117 | 2014/032 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DOLUTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF, INCLUDING DOLUTEGRAVIR SODIUM; REGISTRATION NO/DATE: EU/1/13/892/001-002 20140121 |
| 2673237 | LUC00111 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: SODIUM ZIRCONIUM CYCLOSILICATE; AUTHORISATION NUMBER AND DATE: EU/1/17/1173 20180326 |
| 0370453 | C300359 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ICATIBANT, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, BIJ VOORKEUR ICATIBANTACETAAT; REGISTRATION NO/DATE: EU/1/048/461/001 20080711 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |