Amicus Therap Us Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AMICUS THERAP US, and when can generic versions of AMICUS THERAP US drugs launch?
AMICUS THERAP US has two approved drugs.
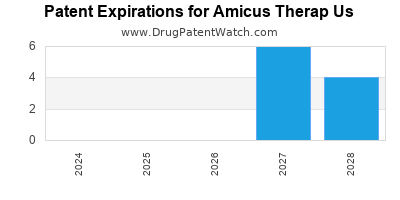
There are sixty-five US patents protecting AMICUS THERAP US drugs.
There are three hundred and five patent family members on AMICUS THERAP US drugs in forty-two countries and eighteen supplementary protection certificates in sixteen countries.
Summary for Amicus Therap Us
| International Patents: | 305 |
| US Patents: | 65 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for Amicus Therap Us
International Patents for Amicus Therap Us Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Eurasian Patent Organization | 038986 | ⤷ Try a Trial |
| Lithuania | 3470077 | ⤷ Try a Trial |
| Cyprus | 1123816 | ⤷ Try a Trial |
| Japan | 2023109807 | ⤷ Try a Trial |
| Taiwan | 202120087 | ⤷ Try a Trial |
| South Korea | 20220146662 | ⤷ Try a Trial |
| Hungary | E064049 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Amicus Therap Us Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2787345 | 122016000090 | Germany | ⤷ Try a Trial | PRODUCT NAME: MIGALASTAT ODER EIN SALZ DAVON, EINSCHLIESSLICH DES HYDROCHLORIDSALZES; REGISTRATION NO/DATE: EU/1/15/1082/001 20160526 |
| 2787345 | PA2016033,C2787345 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: MIGALASTATAS ARBA JO DRUSKA, ISKAITANT IR HIDROCHLORIDO DRUSKA; REGISTRATION NO/DATE: EU/1/15/1082 20160526 |
| 2787345 | CR 2016 00055 | Denmark | ⤷ Try a Trial | PRODUCT NAME: MIGALASTAT ELLER ET SALT HERAF, HERUNDER HYDROGENKLORIDSALTET; REG. NO/DATE: EU/1/15/1082 20160531 |
| 2787345 | 2016/050 | Ireland | ⤷ Try a Trial | PRODUCT NAME: MIGALASTAT OR A SALT THEREOF, INCLUDING THE HYDROCHLORIDE SALT; REGISTRATION NO/DATE: EU/1/15/1082 20160526 |
| 2787345 | PA2016033 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: MIGALASTATAS ARBA JO DRUSKA, ISKAITANT IR HIDROCHLORIDO DRUSKA; REGISTRATION NO/DATE: EU/1/15/1082 20160526 |
| 2787345 | 93312 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: MIGALASTAT OU UN SEL DE CELUI-CI , Y COMPRIS LE SEL DE CHLORHYDRATE; AUTHORISATION NUMBER AND DATE: EU/1/15/1082 - GALAFOLD - MIGALASTAT |
| 2787345 | 132016000116282 | Italy | ⤷ Try a Trial | PRODUCT NAME: MIGALASTAT O UN SUO SALE, COMPRESO IL SALE CLORIDRATO(GALAFOLD); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/15/1082, 20160531 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.