Accord Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ACCORD, and what generic alternatives to ACCORD drugs are available?
ACCORD has one hundred and fifty-five approved drugs.
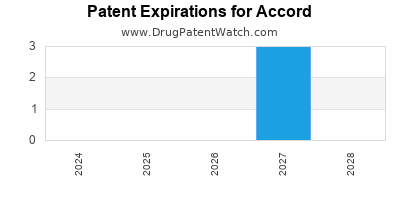
There are four US patents protecting ACCORD drugs. There are three tentative approvals on ACCORD drugs.
There are thirty-five patent family members on ACCORD drugs in eighteen countries and three hundred and eighty-six supplementary protection certificates in eighteen countries.
Summary for Accord
| International Patents: | 35 |
| US Patents: | 4 |
| Tradenames: | 132 |
| Ingredients: | 129 |
| NDAs: | 155 |
| Patent Litigation for Accord: | See patent lawsuits for Accord |
| PTAB Cases with Accord as petitioner: | See PTAB cases with Accord as petitioner |
Drugs and US Patents for Accord
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Accord Hlthcare | SERTRALINE HYDROCHLORIDE | sertraline hydrochloride | TABLET;ORAL | 202825-003 | Nov 7, 2014 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Accord Hlthcare | OLMESARTAN MEDOXOMIL AND HYDROCHLOROTHIAZIDE | hydrochlorothiazide; olmesartan medoxomil | TABLET;ORAL | 209281-002 | Feb 7, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Accord Hlthcare | ISOSORBIDE MONONITRATE | isosorbide mononitrate | TABLET, EXTENDED RELEASE;ORAL | 209684-002 | Oct 24, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Paragraph IV (Patent) Challenges for ACCORD drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
International Patents for Accord Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Israel | 275448 | ⤷ Try a Trial |
| Brazil | PI0706558 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2007084460 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Accord Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1427415 | C 2011 008 | Romania | ⤷ Try a Trial | PRODUCT NAME: APIXABANSI SARURI ACCEPTABILE FARMACEUTIC ALE ACESTUIA; NATIONAL AUTHORISATION NUMBER: RO EU/1/11/691/001, RO EU/1/11/691/002, RO EU/1/11/691/003, RO EU/1/11/691/004, RO EU/1/11/691/005; DATE OF NATIONAL AUTHORISATION: 20110518; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/11/691/001, EU/1/11/691/002, EU/1/11/691/003, EU/1/11/691/004, EU/1/11/691/005; DATE OF FIRST AUTHORISATION IN EEA: 20110518 |
| 0763039 | 122008000023 | Germany | ⤷ Try a Trial | PRODUCT NAME: TEMSIROLIMUS ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/07/424/001 20071119 |
| 0720599 | 92544 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: EZETIMIBE EN COMBINAISON AVEC ROSUVASTATINE OU LEURS SELS PHARMACEUTIQUEMENT ACCEPTEES, Y COMPRIS ROSUVASTATINE SOUD FORME DE SEL DE ZINC; FIRST REGISTRATION DATE: 20140724 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.