CARVEDILOL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Carvedilol, and when can generic versions of Carvedilol launch?
Carvedilol is a drug marketed by ACI, Aurobindo Pharma, Beximco Usa, Chartwell Molecular, Dr Reddys Labs Ltd, Glenmark Generics, Hikma, Lupin, Mylan, Pliva Hrvatska Doo, Rubicon, Sandoz, Sun Pharm Inds Inc, Sun Pharm Inds Ltd, Taro, Teva, Zydus Pharms Usa Inc, Impax Labs Inc, and Sun Pharm Industries. and is included in nineteen NDAs.
The generic ingredient in CARVEDILOL is carvedilol phosphate. There are thirty-seven drug master file entries for this compound. Eight suppliers are listed for this compound. Additional details are available on the carvedilol phosphate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Carvedilol
A generic version of CARVEDILOL was approved as carvedilol phosphate by SUN PHARM INDUSTRIES on October 25th, 2017.
Summary for CARVEDILOL
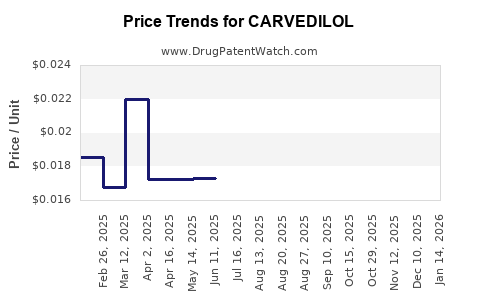
See drug prices for CARVEDILOL
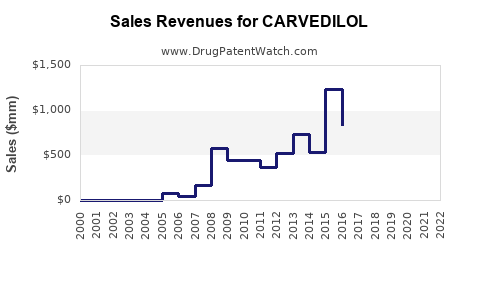
Recent Clinical Trials for CARVEDILOL
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| West China Tianfu Hospital | N/A |
| Chengdu Shangjin Nanfu Hospital | N/A |
| West China Hospital | N/A |
Pharmacology for CARVEDILOL
| Drug Class | alpha-Adrenergic Blocker beta-Adrenergic Blocker |
| Mechanism of Action | Adrenergic alpha-Antagonists Adrenergic beta1-Antagonists Adrenergic beta2-Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for CARVEDILOL
US Patents and Regulatory Information for CARVEDILOL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Impax Labs Inc | CARVEDILOL PHOSPHATE | carvedilol phosphate | CAPSULE, EXTENDED RELEASE;ORAL | 204717-004 | May 7, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Rubicon | CARVEDILOL | carvedilol | TABLET;ORAL | 078165-001 | Sep 5, 2007 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lupin | CARVEDILOL | carvedilol | TABLET;ORAL | 078217-004 | Sep 5, 2007 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


