Eisai Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for EISAI INC, and what generic alternatives to EISAI INC drugs are available?
EISAI INC has eleven approved drugs.
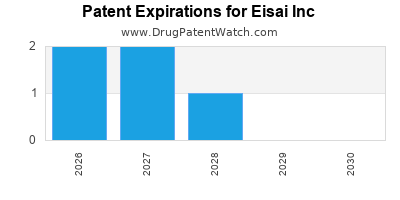
There are twelve US patents protecting EISAI INC drugs.
There are three hundred and ten patent family members on EISAI INC drugs in forty-two countries and thirty-nine supplementary protection certificates in eighteen countries.
Summary for Eisai Inc
| International Patents: | 310 |
| US Patents: | 12 |
| Tradenames: | 8 |
| Ingredients: | 7 |
| NDAs: | 11 |
| Drug Master File Entries: | 1 |
Drugs and US Patents for Eisai Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Eisai Inc | ARICEPT | donepezil hydrochloride | TABLET;ORAL | 020690-001 | Nov 25, 1996 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Eisai Inc | LENVIMA | lenvatinib mesylate | CAPSULE;ORAL | 206947-001 | Feb 13, 2015 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Eisai Inc | LENVIMA | lenvatinib mesylate | CAPSULE;ORAL | 206947-001 | Feb 13, 2015 | RX | Yes | No | 11,090,386 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Eisai Inc | HALAVEN | eribulin mesylate | SOLUTION;INTRAVENOUS | 201532-001 | Nov 15, 2010 | RX | Yes | Yes | RE46965*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Eisai Inc | DAYVIGO | lemborexant | TABLET;ORAL | 212028-001 | Apr 7, 2020 | RX | Yes | No | 8,268,848 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Eisai Inc | LENVIMA | lenvatinib mesylate | CAPSULE;ORAL | 206947-002 | Feb 13, 2015 | RX | Yes | Yes | 7,253,286 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Eisai Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Eisai Inc | LUSEDRA | fospropofol disodium | SOLUTION;INTRAVENOUS | 022244-001 | Dec 12, 2008 | 6,204,257 | ⤷ Try a Trial |
| Eisai Inc | ARICEPT | donepezil hydrochloride | TABLET;ORAL | 020690-001 | Nov 25, 1996 | 5,985,864 | ⤷ Try a Trial |
| Eisai Inc | BANZEL | rufinamide | TABLET;ORAL | 021911-002 | Nov 14, 2008 | 7,750,028*PED | ⤷ Try a Trial |
| Eisai Inc | ARICEPT | donepezil hydrochloride | TABLET;ORAL | 020690-001 | Nov 25, 1996 | 4,895,841 | ⤷ Try a Trial |
| Eisai Inc | BANZEL | rufinamide | SUSPENSION;ORAL | 201367-001 | Mar 3, 2011 | 6,740,669*PED | ⤷ Try a Trial |
| Eisai Inc | HALAVEN | eribulin mesylate | SOLUTION;INTRAVENOUS | 201532-001 | Nov 15, 2010 | 8,097,648 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for EISAI INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | 4 mg and 10 mg | ➤ Subscribe | 2019-02-13 |
| ➤ Subscribe | Tablets | 100 mg, 200 mg and 400 mg | ➤ Subscribe | 2012-11-14 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2010-06-30 |
| ➤ Subscribe | Tablets | 23 mg | ➤ Subscribe | 2013-07-09 |
| ➤ Subscribe | Oral Suspension | 40 mg/mL | ➤ Subscribe | 2014-06-16 |
International Patents for Eisai Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 1698623 | ⤷ Try a Trial |
| Mexico | PA03003362 | ⤷ Try a Trial |
| Russian Federation | 2390354 | ⤷ Try a Trial |
| Canada | 2822994 | ⤷ Try a Trial |
| China | 113683564 | ⤷ Try a Trial |
| New Zealand | 555693 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Eisai Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1087960 | 132011901976051 | Italy | ⤷ Try a Trial | PRODUCT NAME: ERIBULINA(HALAVEN); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/11/678/001-002, 20110317 |
| 0296560 | SPC/GB97/023 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: DONEPEZIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, INCLUDING THE HYDROCHLORIDE; REGISTERED: UK PL 10555/0006 19970214; UK PL 10555/0007 19970214 |
| 1087960 | CA 2011 00022 | Denmark | ⤷ Try a Trial | |
| 0994863 | 91345 | Luxembourg | ⤷ Try a Trial | CERTIFICATE NAME: RUFINAMIDE ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES (INOVELON); FIRST REGISTRATION DATE: 20070116 |
| 1698623 | C20150042 00382 | Estonia | ⤷ Try a Trial | PRODUCT NAME: LENVATINIIB;REG NO/DATE: EU/1/15/1002 01.06.2015 |
| 1415987 | 300764 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LENVATINIB, EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, OF EEN HYDRAAT DAARVAN; REGISTRATION NO/DATE: EU/1/15/1002/001 -002 20150528 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.