Am Regent Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AM REGENT, and what generic alternatives to AM REGENT drugs are available?
AM REGENT has seventy-eight approved drugs.
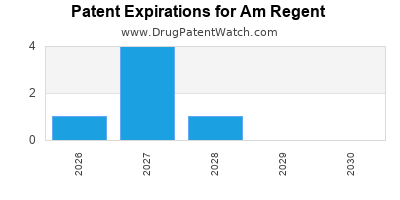
There are seven US patents protecting AM REGENT drugs.
There are sixty-seven patent family members on AM REGENT drugs in thirty-two countries and one hundred and forty-three supplementary protection certificates in fifteen countries.
Drugs and US Patents for Am Regent
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Am Regent | LIDOCAINE HYDROCHLORIDE | lidocaine hydrochloride | INJECTABLE;INJECTION | 080850-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Am Regent | OXALIPLATIN | oxaliplatin | INJECTABLE;INTRAVENOUS | 204378-001 | May 12, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Am Regent | TRALEMENT | cupric sulfate; manganese sulfate; selenious acid; zinc sulfate | SOLUTION;INTRAVENOUS | 209376-001 | Jul 2, 2020 | RX | Yes | Yes | 11,786,548 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Am Regent | CUPRIC SULFATE | cupric sulfate | INJECTABLE;INJECTION | 216324-001 | Dec 16, 2022 | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Am Regent | DOPAMINE HYDROCHLORIDE | dopamine hydrochloride | INJECTABLE;INJECTION | 070826-001 | Feb 11, 1987 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-004 | Feb 4, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Am Regent
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-001 | Jul 25, 2013 | 10,519,252 | ⤷ Try a Trial |
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-004 | Feb 4, 2022 | 11,291,645 | ⤷ Try a Trial |
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-003 | Apr 28, 2021 | 11,291,645 | ⤷ Try a Trial |
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-002 | Oct 8, 2020 | 11,123,321 | ⤷ Try a Trial |
| Am Regent | DEXFERRUM | ferric oxyhydroxide | INJECTABLE;INJECTION | 040024-001 | Feb 23, 1996 | 5,624,668 | ⤷ Try a Trial |
| Am Regent | INJECTAFER | ferric carboxymaltose | SOLUTION;INTRAVENOUS | 203565-001 | Jul 25, 2013 | 11,291,645 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Am Regent Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 100480275 | ⤷ Try a Trial |
| Taiwan | I265028 | ⤷ Try a Trial |
| South Korea | 20180077337 | ⤷ Try a Trial |
| Malaysia | 134394 | ⤷ Try a Trial |
| Germany | 50313653 | ⤷ Try a Trial |
| Lithuania | 1973549 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Am Regent Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0285237 | 95C0008 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL, HEMIHYDRATE; NAT. REGISTRATION NO/DATE: NL 19489 19941107; FIRST REGISTRATION: FR - NL 19489 19941107 |
| 0694547 | 03C0003 | France | ⤷ Try a Trial | PRODUCT NAME: VALGANCICLOVIR AINSI QUE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES ET EN PARTICULIER LE CHLORHYDRATE; NAT. REGISTRATION NO/DATE: NL 27380 20021016; FIRST REGISTRATION: NL - RVG 25 992 20010920 |
| 0122707 | SPC/GB95/031 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: GEMCITABINE, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY- ACCEPTABLE SALT; REGISTERED: NL RVG17854 19950327; UK 00006/0301 19951026; UK 00006/0302 19951026 |
| 1453521 | C201630040 | Spain | ⤷ Try a Trial | PRODUCT NAME: ETINILESTRADIOL Y MEZCLA DE LEVONORGESTREL Y ETINILESTRADIOL; NATIONAL AUTHORISATION NUMBER: 80340; DATE OF AUTHORISATION: 20160122; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): 17/0017/15-S; DATE OF FIRST AUTHORISATION IN EEA: 20150211 |
| 0733366 | SPC/GB98/031 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: 2-BUTYL-4-CHLORO-1-((2'-(1H-TETRAZOL-5-YL) BIPHENYL-4-YL)-METHYL)-5-(HYDROXYMETHYL)IMIDAZOLE (LOSARTAN),OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, IN PARTICULAR A POTASSIUM SALT( LOSARTAN POTASSIUM) AND HYDROCHLOROTHIAZIDE,; REGISTERED: FR 338520.7 19950215; FR 558453.7 19950215; UK 00025/0338 19960412 |
| 1453521 | C 2015 029 | Romania | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL SI ETINILESTRADIOL; NATIONAL AUTHORISATION NUMBER: RO 7793/2015/001; DATE OF NATIONAL AUTHORISATION: 20150612; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): SK. 17/0017/15-S; DATE OF FIRST AUTHORISATION IN EEA: 20150129 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.