ROFLUMILAST Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Roflumilast, and when can generic versions of Roflumilast launch?
Roflumilast is a drug marketed by Alkem Labs Ltd, Aurobindo Pharma Ltd, Breckenridge, Hetero Labs Ltd Iii, Micro Labs, MSN, Mylan, Prinston Inc, Strides Pharma, Torrent, and Zydus Pharms. and is included in eleven NDAs.
The generic ingredient in ROFLUMILAST is roflumilast. There are ten drug master file entries for this compound. Thirteen suppliers are listed for this compound. Additional details are available on the roflumilast profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Roflumilast
A generic version of ROFLUMILAST was approved as roflumilast by MYLAN on July 13th, 2018.
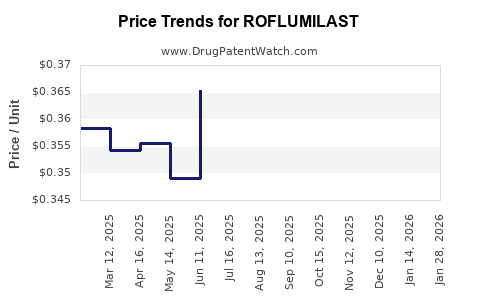
Summary for ROFLUMILAST
Recent Clinical Trials for ROFLUMILAST
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Mansoura University | Phase 1/Phase 2 |
| Jacob Pontoppidan Thyssen | Phase 4 |
| The Novo Nordic Foundation | Phase 4 |
Pharmacology for ROFLUMILAST
| Drug Class | Phosphodiesterase 4 Inhibitor |
| Mechanism of Action | Phosphodiesterase 4 Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ROFLUMILAST
Paragraph IV (Patent) Challenges for ROFLUMILAST
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ZORYVE | Cream | roflumilast | 0.3% | 215985 | 1 | 2023-12-27 |
| DALIRESP | Tablets | roflumilast | 250 mcg | 022522 | 1 | 2019-01-25 |
| DALIRESP | Tablets | roflumilast | 500 mcg | 022522 | 7 | 2015-03-02 |
US Patents and Regulatory Information for ROFLUMILAST
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alkem Labs Ltd | ROFLUMILAST | roflumilast | TABLET;ORAL | 212490-001 | Apr 18, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Zydus Pharms | ROFLUMILAST | roflumilast | TABLET;ORAL | 208303-001 | Feb 10, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Hetero Labs Ltd Iii | ROFLUMILAST | roflumilast | TABLET;ORAL | 208213-001 | Nov 23, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Breckenridge | ROFLUMILAST | roflumilast | TABLET;ORAL | 208236-002 | Oct 19, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Mylan | ROFLUMILAST | roflumilast | TABLET;ORAL | 208257-001 | Jul 13, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Micro Labs | ROFLUMILAST | roflumilast | TABLET;ORAL | 208180-002 | Apr 18, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ROFLUMILAST
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Daxas | roflumilast | EMEA/H/C/001179 Daxas is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment., |
Authorised | no | no | no | 2010-07-05 | |
| AstraZeneca AB | Libertek | roflumilast | EMEA/H/C/002399 Libertek is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment. |
Withdrawn | no | no | no | 2011-02-28 | |
| AstraZeneca AB | Daliresp | roflumilast | EMEA/H/C/002398 Daliresp is indicated for maintenance treatment of severe chronic obstructive pulmonary disease (COPD) (FEV1 post-bronchodilator less than 50% predicted) associated with chronic bronchitis in adult patients with a history of frequent exacerbations as add-on to bronchodilator treatment. |
Withdrawn | no | no | no | 2011-02-28 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |


