PRESTALIA Drug Patent Profile
✉ Email this page to a colleague
When do Prestalia patents expire, and what generic alternatives are available?
Prestalia is a drug marketed by Adhera and is included in one NDA. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has thirty patent family members in twenty-seven countries.
The generic ingredient in PRESTALIA is amlodipine besylate; perindopril arginine. There are fifty drug master file entries for this compound. Additional details are available on the amlodipine besylate; perindopril arginine profile page.
DrugPatentWatch® Generic Entry Outlook for Prestalia
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be October 5, 2029. This may change due to patent challenges or generic licensing.
There is one Paragraph IV patent challenge for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
Summary for PRESTALIA
| International Patents: | 30 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Patent Applications: | 3 |
| Formulation / Manufacturing: | see details |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PRESTALIA |
| What excipients (inactive ingredients) are in PRESTALIA? | PRESTALIA excipients list |
| DailyMed Link: | PRESTALIA at DailyMed |
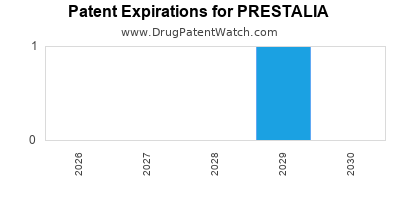
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for PRESTALIA
Generic Entry Date for PRESTALIA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Anatomical Therapeutic Chemical (ATC) Classes for PRESTALIA
Paragraph IV (Patent) Challenges for PRESTALIA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PRESTALIA | Tablets | amlodipine besylate; perindopril arginine | 3.5 mg/2.5 mg, 7 mg/5 mg and 14 mg/10 mg | 205003 | 1 | 2016-11-04 |
US Patents and Regulatory Information for PRESTALIA
PRESTALIA is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of PRESTALIA is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting PRESTALIA
.alpha. crystalline form of the arginine salt of perindopril, a process for its preparation and pharmaceutical compositions containing it
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HYPERTENSION
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-001 | Jan 21, 2015 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-002 | Jan 21, 2015 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-003 | Jan 21, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for PRESTALIA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-003 | Jan 21, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-001 | Jan 21, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-002 | Jan 21, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for PRESTALIA
When does loss-of-exclusivity occur for PRESTALIA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 07220435
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0708278
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 44467
Estimated Expiration: ⤷ Try a Trial
China
Patent: 1389603
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0160644
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 17753
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 89182
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 4716
Estimated Expiration: ⤷ Try a Trial
Patent: 0801777
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 89182
Estimated Expiration: ⤷ Try a Trial
France
Patent: 97866
Estimated Expiration: ⤷ Try a Trial
Georgia, Republic of
Patent: 0125433
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 29669
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 27898
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 55454
Estimated Expiration: ⤷ Try a Trial
Patent: 09534295
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 1035
Estimated Expiration: ⤷ Try a Trial
Montenegro
Patent: 456
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 276
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 0367
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 083535
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 89182
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 754
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 89182
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 0807024
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 080106948
Estimated Expiration: ⤷ Try a Trial
Patent: 120001818
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 81982
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 905
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering PRESTALIA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Poland | 359321 | Nowa sól perindoprilu i zawierające ją kompozycjefarmaceutyczne (NEW SALT PERINDOPRYL AND PHARMACEUTICAL COMPOSUNDS CONTAINING IT) | ⤷ Try a Trial |
| Norway | 324323 | ⤷ Try a Trial | |
| Austria | 271036 | ⤷ Try a Trial | |
| China | 101389603 | Crystalline form of the arginine salt of perindopril, process for preparing it, and pharmaceutical compositions comprising it | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PRESTALIA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0443983 | 2007C/043 | Belgium | ⤷ Try a Trial | PRODUCT NAME: AMLODIPINE ET VALSARTAN; NATL. REGISTRATION NO/DATE: EU/1/06/370/001 20070118; FIRST REGISTRATION: CH 57771 20061222 |
| 0503785 | CA 2011 00026 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF OLMESARTAN MEDOXOMIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND AMLODIPINE BESYLATE AND HYDROCHLOROTHIAZIDE; NAT. REG. NO/DATE: 46260-46269 (DK) 20110323; FIRST REG. NO/DATE: DE 79810.00.00 20101216 |
| 0443983 | C00443983/03 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VALSARTAN + AMLODIPINE + HYDROCHLOROTHIAZIDE; REGISTRATION NUMBER/DATE: SWISSMEDIC 59407 16.09.2009 |
| 0503785 | C300375 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN OLMESARTAN MEDOXOMIL, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT EN AMLODIPINEBESILAAT; REGISTRATION NO/DATE: RVG100984, RVG100986-87, RVG100989-91, RVG100993-95 20080819 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


