NORETHINDRONE Drug Patent Profile
✉ Email this page to a colleague
When do Norethindrone patents expire, and when can generic versions of Norethindrone launch?
Norethindrone is a drug marketed by Amneal Pharms, Glenmark Generics, Lupin Ltd, Mylan Labs Ltd, Naari Pte Ltd, Novast Labs, Aurobindo Pharma Ltd, Barr, Barr Labs Inc, Glenmark Pharms Ltd, Dr Reddys Labs Sa, Mylan, Apotex, Xiromed, Watson Labs, Watson Pharms Teva, Watson Labs Teva, and Apil. and is included in sixty-three NDAs.
The generic ingredient in NORETHINDRONE is mestranol; norethindrone. There are eleven drug master file entries for this compound. Additional details are available on the mestranol; norethindrone profile page.
Summary for NORETHINDRONE
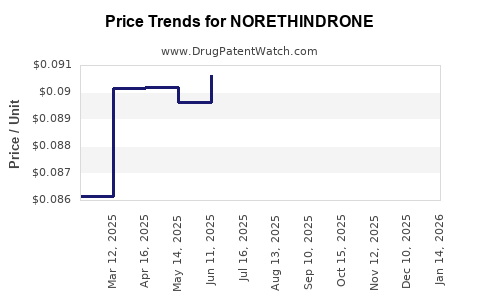
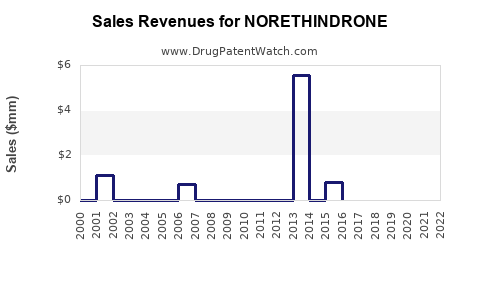
See drug prices for NORETHINDRONE
Recent Clinical Trials for NORETHINDRONE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Bio Genuine (Shanghai) Biotech Co., Ltd. | Phase 3 |
| Myovant Sciences GmbH | Phase 4 |
| University of Chicago | Phase 4 |
Pharmacology for NORETHINDRONE
| Drug Class | Progestin |
Medical Subject Heading (MeSH) Categories for NORETHINDRONE
US Patents and Regulatory Information for NORETHINDRONE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Watson Pharms Teva | NORETHINDRONE AND ETHINYL ESTRADIOL | ethinyl estradiol; norethindrone | TABLET;ORAL-21 | 070685-001 | Jan 29, 1987 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Amneal Pharms | NORETHINDRONE ACETATE AND ETHINYL ESTRADIOL AND FERROUS FUMARATE | ethinyl estradiol; norethindrone acetate | TABLET;ORAL | 207514-001 | Sep 11, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Watson Labs | NORETHINDRONE AND ETHINYL ESTRADIOL | ethinyl estradiol; norethindrone | TABLET;ORAL-28 | 078323-001 | Feb 4, 2010 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


