INFUVITE ADULT Drug Patent Profile
✉ Email this page to a colleague
When do Infuvite Adult patents expire, and what generic alternatives are available?
Infuvite Adult is a drug marketed by Sandoz Canada Inc and is included in one NDA.
The generic ingredient in INFUVITE ADULT is alpha-tocopherol acetate; ascorbic acid; biotin; cholecalciferol; cyanocobalamin; dexpanthenol; folic acid; niacinamide; pyridoxine hydrochloride; riboflavin 5'-phosphate sodium; thiamine hydrochloride; vitamin a palmitate; vitamin k. There are two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the alpha-tocopherol acetate; ascorbic acid; biotin; cholecalciferol; cyanocobalamin; dexpanthenol; folic acid; niacinamide; pyridoxine hydrochloride; riboflavin 5'-phosphate sodium; thiamine hydrochloride; vitamin a palmitate; vitamin k profile page.
Summary for INFUVITE ADULT
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Formulation / Manufacturing: | see details |
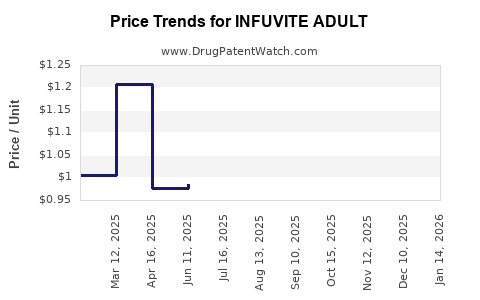
| Drug Prices: | Drug price information for INFUVITE ADULT |
| DailyMed Link: | INFUVITE ADULT at DailyMed |
Pharmacology for INFUVITE ADULT
| Ingredient-type | Analogs/Derivatives Vitamin B 6 |
| Drug Class | Vitamin A Vitamin B12 Vitamin B6 Analog Vitamin C Vitamin D Vitamin K Warfarin Reversal Agent |
| Physiological Effect | Increased Prothrombin Activity Reversed Anticoagulation Activity |
Anatomical Therapeutic Chemical (ATC) Classes for INFUVITE ADULT
US Patents and Regulatory Information for INFUVITE ADULT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sandoz Canada Inc | INFUVITE ADULT | alpha-tocopherol acetate; ascorbic acid; biotin; cholecalciferol; cyanocobalamin; dexpanthenol; folic acid; niacinamide; pyridoxine hydrochloride; riboflavin 5'-phosphate sodium; thiamine hydrochloride; vitamin a palmitate; vitamin k | SOLUTION;INTRAVENOUS | 021163-001 | May 18, 2000 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Sandoz Canada Inc | INFUVITE ADULT | alpha-tocopherol acetate; ascorbic acid; biotin; cholecalciferol; cyanocobalamin; dexpanthenol; folic acid; niacinamide; pyridoxine hydrochloride; riboflavin 5'-phosphate sodium; thiamine hydrochloride; vitamin a palmitate; vitamin k | SOLUTION;INTRAVENOUS | 021163-002 | Jun 16, 2003 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


