Icosapent ethyl - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for icosapent ethyl and what is the scope of freedom to operate?
Icosapent ethyl
is the generic ingredient in two branded drugs marketed by Apotex, Ascent Pharms Inc, Dr Reddys, Hikma, Humanwell Puracap, Strides Pharma, Teva Pharms Usa, Zydus, and Amarin Pharms, and is included in nine NDAs. There are seventy patents protecting this compound and three Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Icosapent ethyl has three hundred and forty-nine patent family members in forty-five countries.
There are six drug master file entries for icosapent ethyl. Fifteen suppliers are listed for this compound.
Summary for icosapent ethyl
| International Patents: | 349 |
| US Patents: | 70 |
| Tradenames: | 2 |
| Applicants: | 9 |
| NDAs: | 9 |
| Drug Master File Entries: | 6 |
| Finished Product Suppliers / Packagers: | 15 |
| Raw Ingredient (Bulk) Api Vendors: | 60 |
| Clinical Trials: | 17 |
| Patent Applications: | 2,462 |
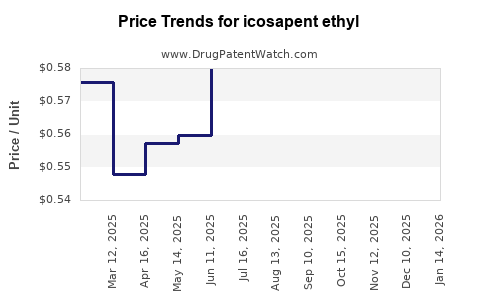
| Drug Prices: | Drug price trends for icosapent ethyl |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for icosapent ethyl |
| What excipients (inactive ingredients) are in icosapent ethyl? | icosapent ethyl excipients list |
| DailyMed Link: | icosapent ethyl at DailyMed |
Recent Clinical Trials for icosapent ethyl
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Cancer Institute (NCI) | Phase 1/Phase 2 |
| Canadian Medical and Surgical Knowledge Translation Research Group | Phase 4 |
| St. Michael's Hospital, Toronto | Phase 4 |
Paragraph IV (Patent) Challenges for ICOSAPENT ETHYL
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| VASCEPA | Capsules | icosapent ethyl | 500 mg | 202057 | 1 | 2017-08-29 |
| VASCEPA | Capsules | icosapent ethyl | 1 g | 202057 | 4 | 2016-07-26 |
US Patents and Regulatory Information for icosapent ethyl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-001 | Jul 26, 2012 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-001 | Jul 26, 2012 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for icosapent ethyl
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-001 | Jul 26, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for icosapent ethyl
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Amarin Pharmaceuticals Ireland Limited | Vazkepa | icosapent ethyl | EMEA/H/C/005398 Indicated to reduce cardiovascular risk as an adjunct to statin therapy. |
Authorised | no | no | no | 2021-03-26 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for icosapent ethyl
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Russian Federation | 2538691 | СТАБИЛЬНЫЕ ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ И СПОСОБЫ ИХ ПРИМЕНЕНИЯ (STABLE PHARMACEUTICAL COMPOSITIONS AND METHODS FOR USING THEM) | ⤷ Try a Trial |
| Russian Federation | 2648826 | ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ЕРА И СЕРДЕЧНО-СОСУДИСТОЕ СРЕДСТВО, И СПОСОБЫ ИХ ПРИМЕНЕНИЯ (PHARMACEUTICAL COMPOSITIONS CONTAINING EPA AND CARDIOVASCULAR AGENT AND APPLICATION METHODS THEREOF) | ⤷ Try a Trial |
| South Korea | 20130137055 | COMPOSITION FOR PREVENTION OF OCCURRENCE OF CARDIOVASCULAR EVENT | ⤷ Try a Trial |
| Canada | 3026006 | COMPOSITIONS ET PROCEDES POUR ABAISSER LES TRIGLYCERIDES SANS ELEVER LES TAUX DE LDL-C CHEZ UN SUJET SOUMIS A UNE THERAPIE CONCOMITANTE AUX STATINES (COMPOSITIONS AND METHODS FOR LOWERING TRIGLYCERIDES WITHOUT RAISING LDL-C LEVELS IN A SUBJECT ON CONCOMITANT STATIN THERAPY) | ⤷ Try a Trial |
| Cyprus | 1120102 | ⤷ Try a Trial | |
| World Intellectual Property Organization (WIPO) | 2010147994 | ⤷ Try a Trial | |
| Australia | 2023200784 | Methods of reducing the risk of cardiovascular events in a subject | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for icosapent ethyl
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2022495 | 122021000058 | Germany | ⤷ Try a Trial | PRODUCT NAME: ICOSAPENT-ETHYL; REGISTRATION NO/DATE: EU/1/20/1524 20210326 |
| 2443246 | 122021000056 | Germany | ⤷ Try a Trial | PRODUCT NAME: VAZKEPA (ICOSAPENT ETHYL); REGISTRATION NO/DATE: EU/1/20/1524 20210326 |
| 2443246 | PA2021522 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: IKOZAPENTO ETILAS; REGISTRATION NO/DATE: EU/1/20/1524 20210326 |
| 2022495 | C202130051 | Spain | ⤷ Try a Trial | PRODUCT NAME: ICOSAPENTO DE ETILO; NATIONAL AUTHORISATION NUMBER: EU/1/20/1524; DATE OF AUTHORISATION: 20210326; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1524; DATE OF FIRST AUTHORISATION IN EEA: 20210326 |
| 2443246 | 39/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: ICOSAPENT-ETHYL (= EICOSAPENTAENSAEUREETHYLESTER,; REGISTRATION NO/DATE: EU/1/20/1524 20210329 |
| 2443246 | 2021C/538 | Belgium | ⤷ Try a Trial | PRODUCT NAME: VAZKEPA - ICOSAPENT ETHYL; AUTHORISATION NUMBER AND DATE: EU/1/20/1524 20210329 |
| 2443246 | C20210022 00395 | Estonia | ⤷ Try a Trial | PRODUCT NAME: IKOSAPENTETUEUEL;REG NO/DATE: EU/1/20/1524 29.03.2021 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |