Radius Company Profile
✉ Email this page to a colleague
What is the competitive landscape for RADIUS, and what generic alternatives to RADIUS drugs are available?
RADIUS has two approved drugs.
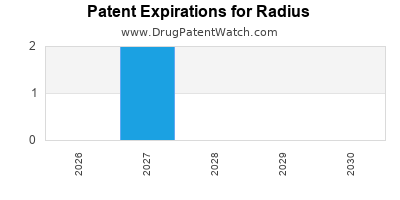
There are nine US patents protecting RADIUS drugs.
There are sixty-three patent family members on RADIUS drugs in thirty-one countries and seven supplementary protection certificates in six countries.
Drugs and US Patents for Radius
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | RE49444 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | 11,255,842 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | 8,148,333 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Radius | BINOSTO | alendronate sodium | TABLET, EFFERVESCENT;ORAL | 202344-001 | Mar 12, 2012 | RX | Yes | Yes | 9,592,195 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Radius
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Radius | BINOSTO | alendronate sodium | TABLET, EFFERVESCENT;ORAL | 202344-001 | Mar 12, 2012 | 7,488,496 | ⤷ Try a Trial |
| Radius | BINOSTO | alendronate sodium | TABLET, EFFERVESCENT;ORAL | 202344-001 | Mar 12, 2012 | 7,964,212 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Radius Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 2021007056 | ⤷ Try a Trial |
| Cyprus | 1119198 | ⤷ Try a Trial |
| Russian Federation | 2013109765 | ⤷ Try a Trial |
| South Korea | 20150020289 | ⤷ Try a Trial |
| South Korea | 101512377 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Radius Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2073789 | 122023000031 | Germany | ⤷ Try a Trial | PRODUCT NAME: ABALOPARATID; REGISTRATION NO/DATE: EU/1/22/1706 20221212 |
| 1175904 | 2007C/048 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ALENDRONATE DE SODIUM/COLECALCIFEROL; AUTHORISATION NUMBER AND DATE: EU/1/05/310/001 20050826 |
| 2073789 | CA 2023 00019 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ABALOPARATID; REG. NO/DATE: EU/1/22/1706 20221213 |
| 2073789 | 2023C/523 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ABALOPARATIDE; AUTHORISATION NUMBER AND DATE: EU/1/22/1706 20221213 |
| 2073789 | 23/2023 | Austria | ⤷ Try a Trial | PRODUCT NAME: ABALOPARATID; REGISTRATION NO/DATE: EU/1/22/1706 (MITTEILUNG) 20221213 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.