CEPHALEXIN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cephalexin, and what generic alternatives are available?
Cephalexin is a drug marketed by Alkem Labs Ltd, Anda Repository, Apothecon, Aurobindo Pharma, Barr, Belcher Pharms, Chartwell Rx, Facta Farma, Hikma, Ivax Sub Teva Pharms, Lupin, Purepac Pharm, Stevens J, Sun Pharm Inds (in), Sun Pharm Inds Ltd, Teva, Yoshitomi, Hikma Pharms, Vitarine, and Yung Shin Pharm. and is included in fifty NDAs.
The generic ingredient in CEPHALEXIN is cephalexin. There are twenty-nine drug master file entries for this compound. Thirty-three suppliers are listed for this compound. Additional details are available on the cephalexin profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Cephalexin
A generic version of CEPHALEXIN was approved as cephalexin by TEVA on February 13th, 1987.
Summary for CEPHALEXIN
| US Patents: | 0 |
| Applicants: | 20 |
| NDAs: | 50 |
| Finished Product Suppliers / Packagers: | 33 |
| Raw Ingredient (Bulk) Api Vendors: | 68 |
| Clinical Trials: | 61 |
| Patent Applications: | 4,165 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for CEPHALEXIN |
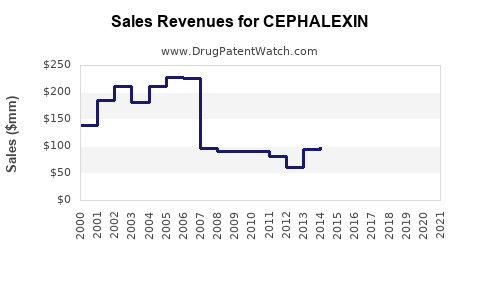
| Drug Sales Revenues: | Drug sales revenues for CEPHALEXIN |
| What excipients (inactive ingredients) are in CEPHALEXIN? | CEPHALEXIN excipients list |
| DailyMed Link: | CEPHALEXIN at DailyMed |
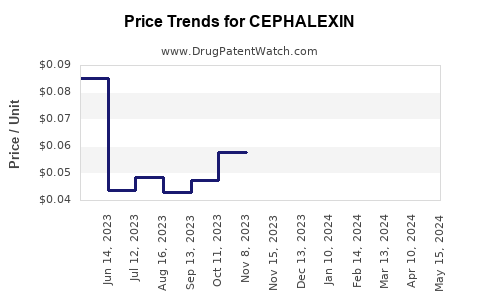
See drug prices for CEPHALEXIN
Recent Clinical Trials for CEPHALEXIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Network of Canadian Emergency Researchers (NCER) | Phase 4 |
| Centre Of Research Excellence in Tuberculosis Control | Phase 2 |
| Western Sydney Local Health District | Phase 2 |
Pharmacology for CEPHALEXIN
| Drug Class | Cephalosporin Antibacterial |
Medical Subject Heading (MeSH) Categories for CEPHALEXIN
US Patents and Regulatory Information for CEPHALEXIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Yung Shin Pharm | CEPHALEXIN | cephalexin | FOR SUSPENSION;ORAL | 065336-002 | Jul 25, 2007 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Teva | CEPHALEXIN | cephalexin | CAPSULE;ORAL | 062702-002 | Feb 13, 1987 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Stevens J | CEPHALEXIN | cephalexin | CAPSULE;ORAL | 062870-001 | Mar 17, 1988 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


