Lupin Company Profile
✉ Email this page to a colleague
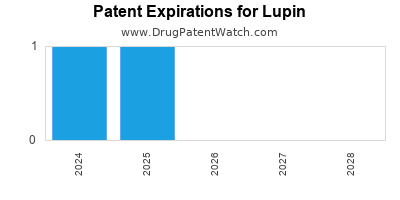
What is the competitive landscape for LUPIN, and when can generic versions of LUPIN drugs launch?
LUPIN has two hundred and thirty-one approved drugs.
There are nineteen US patents protecting LUPIN drugs. There are twenty-two tentative approvals on LUPIN drugs.
There are ninety-nine patent family members on LUPIN drugs in twenty-four countries and six hundred and seventy-eight supplementary protection certificates in seventeen countries.
Summary for Lupin
| International Patents: | 99 |
| US Patents: | 19 |
| Tradenames: | 189 |
| Ingredients: | 169 |
| NDAs: | 231 |
| Patent Litigation for Lupin: | See patent lawsuits for Lupin |
| PTAB Cases with Lupin as petitioner: | See PTAB cases with Lupin as petitioner |
Drugs and US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin Ltd | DIVALPROEX SODIUM | divalproex sodium | TABLET, EXTENDED RELEASE;ORAL | 209286-002 | Oct 18, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Pharms | AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE | amlodipine besylate; benazepril hydrochloride | CAPSULE;ORAL | 078466-005 | Jul 5, 2011 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | FYAVOLV | ethinyl estradiol; norethindrone acetate | TABLET;ORAL | 204213-001 | Dec 10, 2015 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | CELECOXIB | celecoxib | CAPSULE;ORAL | 202240-004 | Jun 9, 2015 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,472,563 | ⤷ Try a Trial |
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,068,833 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,844,002 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,760,090 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for LUPIN drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Inhalation Solution | Eq. 0.015 mg base/2 mL | ➤ Subscribe | 2009-10-01 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Oral Suspension | 500 mg/5 mL | ➤ Subscribe | 2014-07-22 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
International Patents for Lupin Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2626648 | ⤷ Try a Trial |
| Japan | 6276828 | ⤷ Try a Trial |
| Japan | 2015110671 | ⤷ Try a Trial |
| Japan | 2021042246 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Lupin Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1453521 | 132016000025143 | Italy | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL ED ETINILESTRADIOLO(SEASONIQUE); AUTHORISATION NUMBER(S) AND DATE(S): 17/0017/15-S, 20150211;042139016, 20150414 |
| 0600675 | 10C0036 | France | ⤷ Try a Trial | PRODUCT NAME: SILODOSIN; REGISTRATION NO/DATE IN FRANCE: EU/1/09/608/001 DU 20100129; REGISTRATION NO/DATE AT EEC: EU/1/09/608/001 DU 20100129 |
| 2316456 | C 2017 047 | Romania | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE DE NALTREXONA SAU O SARE ACCEPTABILA FARMACEUTIC A ACESTEIA, IN PARTICULAR CLORHIDRAT DE NALTREXONA SI BUPROPION SAU O SARE ACCEPTABILA FARMACEUTIC A ACESTUIA, IN PARTICULAR CLORHIDRAT DE BUPROPION; NATIONAL AUTHORISATION NUMBER: EU/1/14/988; DATE OF NATIONAL AUTHORISATION: 20150326; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/14/988; DATE OF FIRST AUTHORISATION IN EEA: 20150326 |
| 2435024 | 15/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: FORMOTEROLFUMARAT-DIHYDRAT / GLYCOPYRRONIUMBROMID / BUDESONID; REGISTRATION NO/DATE: EU/1/20/1498 (MITTEILUNG) 20201210 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.