vizimpro Drug Patent Profile
✉ Email this page to a colleague
When do Vizimpro patents expire, and what generic alternatives are available?
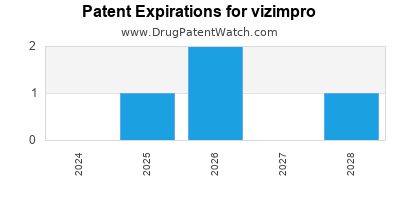
Vizimpro is a drug marketed by Pfizer and is included in one NDA. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this compound. Additional details are available on the dacomitinib profile page.
DrugPatentWatch® Generic Entry Outlook for Vizimpro
Vizimpro was eligible for patent challenges on September 27, 2022.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be August 26, 2028. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for vizimpro
| International Patents: | 93 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 29 |
| Clinical Trials: | 5 |
| Patent Applications: | 204 |
| Drug Prices: | Drug price information for vizimpro |
| What excipients (inactive ingredients) are in vizimpro? | vizimpro excipients list |
| DailyMed Link: | vizimpro at DailyMed |

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for vizimpro
Generic Entry Date for vizimpro*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for vizimpro
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Samsung Medical Center | Phase 2 |
| Cancer Institute and Hospital, Chinese Academy of Medical Sciences | Phase 2 |
| Fondazione Ricerca Traslazionale | Phase 2 |
Anatomical Therapeutic Chemical (ATC) Classes for vizimpro
US Patents and Regulatory Information for vizimpro
vizimpro is protected by four US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of vizimpro is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting vizimpro
Method for treating gefitinib resistant cancer
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: ADMINISTERING DAILY A UNIT DOSAGE OF AN IRREVERSIBLE EGFR INHIBITOR COVALENTLY BINDING AS CLAIMED FOR 1ST LINE TREATMENT OF GEFITINIB OR ERLOTINIB RESISTANT METASTATIC NSCLC WITH EGFR EXON 19 DELETION OR EXON 21 L858R SUBSTITUTION WITH T790M MUTATION
Method for treating gefitinib resistant cancer
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: ADMINISTERING DAILY A UNIT DOSAGE OF AN IRREVERSIBLE EGFR INHIBITOR COVALENTLY BINDING AS CLAIMED FOR 1ST LINE TREATMENT OF GEFITINIB OF ERLOTINIB RESISTANT METASTATIC NSCLC WITH EGFR EXON 19 DELETION OR EXON 21 L858R SUBSTITUTION
4-phenylamino-quinazolin-6-yl-amides
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
4-phenylamino-quinazolin-6-yl-amides
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: FIRST-LINE TREATMENT OF METASTATIC NON SMALL-CELL LUNG CANCER (NSCLC) WITH EGFR EXON 19 DELETIONS OR EXON 21 (L858R) SUBSTITUTION MUTATIONS AS DETECTED BY AN FDA-APPROVED TEST
FDA Regulatory Exclusivity protecting vizimpro
FIRST-LINE TREATMENT OF PATIENTS WITH METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC) WITH EPIDERMAL GROWTH FACTOR RECEPTOR (EGFR) EXON 19 DELETION OR EXON 21 L858R SUBSTITUTION MUTATIONS AS DETECTED BY AN FDA-APPROVED TEST
Exclusivity Expiration: ⤷ Try a Trial
INDICATED FOR THE FIRST-LINE TREATMENT OF PATIENTS WITH METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC) WITH EPIDERMAL GROWTH FACTOR (EGFR) EXON 19 DELETION OR EXON 21 L858R SUBSTITUTION MUTATIONS AS DETECTED BY AN FDA-APPROVED TEST
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-001 | Sep 27, 2018 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-002 | Sep 27, 2018 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-001 | Sep 27, 2018 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-002 | Sep 27, 2018 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-003 | Sep 27, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Pfizer | VIZIMPRO | dacomitinib | TABLET;ORAL | 211288-003 | Sep 27, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for vizimpro
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pfizer Europe MA EEIG | Vizimpro | dacomitinib | EMEA/H/C/004779 Vizimpro, as monotherapy, is indicated for the first-line treatment of adult patients with locally advanced or metastatic non small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) activating mutations. |
Authorised | no | no | no | 2019-04-02 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for vizimpro
See the table below for patents covering vizimpro around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Russian Federation | 2007132902 | СПОСОБ ЛЕЧЕНИЯ РАКА, УСТОЙЧИВОГО К ГЕФИТИНИБУ | ⤷ Try a Trial |
| Brazil | PI0606839 | uso de uma composição farmacêutica compreendendo um inibidor irreversìvel de receptor de fator de crescimento epidérmico (egfr) | ⤷ Try a Trial |
| Norway | 339692 | ⤷ Try a Trial | |
| China | 108421044 | 治疗吉非替尼耐药性癌症的方法 (METHOD FOR TREATING GEFITINIB RESISTANT CANCER) | ⤷ Try a Trial |
| European Patent Office | 1746999 | 4-PHENYLAMINO-QUINAZOLIN-6-YL-AMIDES (4-PHENYLAMINO-QUINAZOLIN-6-YL-AMIDES) | ⤷ Try a Trial |
| Netherlands | 1028967 | 4-Fenylamino-chinazoline-6-yl-amiden. | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for vizimpro
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1746999 | 1990044-8 | Sweden | ⤷ Try a Trial | PRODUCT NAME: DACOMITINIB OR APHARMACEUTICAL ACCEPTABLE SALT THEROF; REG. NO/DATE: EU/1/19/1354 20190404 |
| 1746999 | LUC00127 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: DACOMITINIB ET SES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES (VIZIMPRO ); AUTHORISATION NUMBER AND DATE: EU/1/19/1354 20190404 |
| 1746999 | C201930055 | Spain | ⤷ Try a Trial | PRODUCT NAME: DACOMITINIB O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/19/1354; DATE OF AUTHORISATION: 20190402; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/19/1354; DATE OF FIRST AUTHORISATION IN EEA: 20190402 |
| 1746999 | C20190030 00298 | Estonia | ⤷ Try a Trial | PRODUCT NAME: DAKOMITINIIB;REG NO/DATE: EU/1/19/1354 04.04.2019 |
| 1848414 | C 2016 026 | Romania | ⤷ Try a Trial | PRODUCT NAME: OSIMERTINIB; NATIONAL AUTHORISATION NUMBER: EU/1/16/1086; DATE OF NATIONAL AUTHORISATION: 20160202; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/16/1086; DATE OF FIRST AUTHORISATION IN EEA: 20160202 |
| 1746999 | 43/2019 | Austria | ⤷ Try a Trial | PRODUCT NAME: DACOMITINIB ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON, INSBESONDERE DACOMITINIBMONOHYDRAT; REGISTRATION NO/DATE: EU/1/19/1354 (MITTEILUNG) 20190404 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


