caprelsa Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Caprelsa, and when can generic versions of Caprelsa launch?
Caprelsa is a drug marketed by Genzyme Corp and is included in one NDA. There is one patent protecting this drug.
This drug has forty patent family members in thirty-three countries.
The generic ingredient in CAPRELSA is vandetanib. One supplier is listed for this compound. Additional details are available on the vandetanib profile page.
DrugPatentWatch® Generic Entry Outlook for Caprelsa
Caprelsa was eligible for patent challenges on April 6, 2015.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be August 8, 2028. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for caprelsa
| International Patents: | 40 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 137 |
| Clinical Trials: | 17 |
| Patent Applications: | 5,319 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for caprelsa |
| What excipients (inactive ingredients) are in caprelsa? | caprelsa excipients list |
| DailyMed Link: | caprelsa at DailyMed |
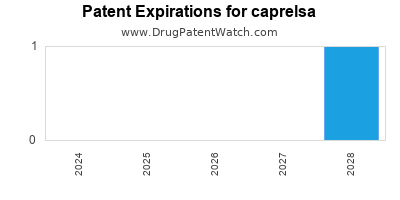

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for caprelsa
Generic Entry Date for caprelsa*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for caprelsa
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Samsung Medical Center | Phase 2 |
| AstraZeneca | Phase 2 |
| AstraZeneca | Phase 1 |
Pharmacology for caprelsa
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Organic Cation Transporter 2 Inhibitors P-Glycoprotein Inhibitors Protein Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for caprelsa
US Patents and Regulatory Information for caprelsa
caprelsa is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of caprelsa is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting caprelsa
Pharmaceutical compositions comprising ZD6474
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-001 | Apr 6, 2011 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-002 | Apr 6, 2011 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for caprelsa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-002 | Apr 6, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-001 | Apr 6, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-001 | Apr 6, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-001 | Apr 6, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Genzyme Corp | CAPRELSA | vandetanib | TABLET;ORAL | 022405-002 | Apr 6, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for caprelsa
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Sanofi B.V. | Caprelsa | vandetanib | EMEA/H/C/002315 Caprelsa is indicated for the treatment of aggressive and symptomatic medullary thyroid cancer (MTC) in patients with unresectable locally advanced or metastatic disease.Caprelsa is indicated in adults, children and adolescents aged 5 years and older.For patients in whom re-arranged-during-transfection(RET) mutation is not known or is negative, a possible lower benefit should be taken into account before individual treatment decision. |
Authorised | no | no | no | 2012-02-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for caprelsa
When does loss-of-exclusivity occur for caprelsa?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9059
Estimated Expiration: ⤷ Try a Trial
Patent: 0045
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 05244650
Estimated Expiration: ⤷ Try a Trial
Austria
Patent: 39841
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0511253
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 65513
Estimated Expiration: ⤷ Try a Trial
China
Patent: 1031303
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0090547
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 09500
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 53431
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 53431
Estimated Expiration: ⤷ Try a Trial
Germany
Patent: 2005016105
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 99220
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 8928
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 40947
Estimated Expiration: ⤷ Try a Trial
Patent: 07538060
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 8868
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 06013501
Estimated Expiration: ⤷ Try a Trial
Montenegro
Patent: 505
Estimated Expiration: ⤷ Try a Trial
Patent: 778
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 0931
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 8324
Estimated Expiration: ⤷ Try a Trial
Patent: 065735
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 53431
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 53431
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 83343
Estimated Expiration: ⤷ Try a Trial
Patent: 06145357
Estimated Expiration: ⤷ Try a Trial
Saudi Arabia
Patent: 260133
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 112
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 53431
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 0609110
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1192935
Estimated Expiration: ⤷ Try a Trial
Patent: 070034499
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 30025
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 55269
Estimated Expiration: ⤷ Try a Trial
Patent: 0612953
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 732
Estimated Expiration: ⤷ Try a Trial
United Kingdom
Patent: 11378
Estimated Expiration: ⤷ Try a Trial
Uruguay
Patent: 901
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering caprelsa around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Poland | 1753431 | ⤷ Try a Trial | |
| South Korea | 100618065 | ⤷ Try a Trial | |
| Chile | 2004001178 | ⤷ Try a Trial | |
| Australia | 729968 | ⤷ Try a Trial | |
| Japan | 2003513089 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for caprelsa
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1244647 | C01244647/01 | Switzerland | ⤷ Try a Trial | FORMER OWNER: ASTRAZENECA AB, SE |
| 1244647 | CA 2012 00032 | Denmark | ⤷ Try a Trial | |
| 1244647 | C300543 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: VANDETANIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/11/749/001-002 20120217 |
| 1244647 | C 2012 023 | Romania | ⤷ Try a Trial | PRODUCT NAME: VANDETANIB SAU O SARE ACCEPTABILA FARMACEUTIC AACESTUIA; NATIONAL AUTHORISATION NUMBER: RO EU/1/11/749/001, RO EU/1/11/749/002; DATE OF NATIONAL AUTHORISATION: 20120217; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EMEA EU/1/11/749/001, EMEA EU/1/11/749/002; DATE OF FIRST AUTHORISATION IN EEA: 20120217 |
| 1244647 | 300543 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: VANDETANIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/11/749/001-002 20120217 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


