BYDUREON BCISE Drug Patent Profile
✉ Email this page to a colleague
When do Bydureon Bcise patents expire, and what generic alternatives are available?
Bydureon Bcise is a drug marketed by Astrazeneca Ab and is included in one NDA. There are thirteen patents protecting this drug.
This drug has four hundred and five patent family members in forty-eight countries.
The generic ingredient in BYDUREON BCISE is exenatide synthetic. There are seven drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the exenatide synthetic profile page.
DrugPatentWatch® Generic Entry Outlook for Bydureon Bcise
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 4, 2031. This may change due to patent challenges or generic licensing.
There have been eight patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for BYDUREON BCISE
| International Patents: | 405 |
| US Patents: | 13 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 39 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for BYDUREON BCISE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BYDUREON BCISE |
| What excipients (inactive ingredients) are in BYDUREON BCISE? | BYDUREON BCISE excipients list |
| DailyMed Link: | BYDUREON BCISE at DailyMed |
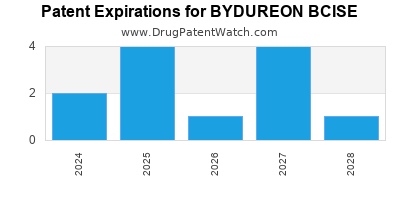

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for BYDUREON BCISE
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for BYDUREON BCISE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| The University of Texas Health Science Center, Houston | Phase 2 |
| University of Washington | Phase 3 |
| Dasman Diabetes Institute | Phase 4 |
Pharmacology for BYDUREON BCISE
| Drug Class | GLP-1 Receptor Agonist |
| Mechanism of Action | Glucagon-like Peptide-1 (GLP-1) Agonists |
Anatomical Therapeutic Chemical (ATC) Classes for BYDUREON BCISE
US Patents and Regulatory Information for BYDUREON BCISE
BYDUREON BCISE is protected by thirteen US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of BYDUREON BCISE is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting BYDUREON BCISE
C-aryl glucoside SGLT2 inhibitors and method
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Methods for treating diabetes and reducing body weight
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Crystal structures of SGLT2 inhibitors and processes for preparing same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Sustained release formulations using non-aqueous carriers
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Method for treating diabetes
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Polymer-based sustained release device
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Methods for treating diabetes and reducing body weight
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
FDA Regulatory Exclusivity protecting BYDUREON BCISE
NEW PATIENT POPULATION
Exclusivity Expiration: ⤷ Sign Up
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for BYDUREON BCISE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | BYDUREON BCISE | exenatide synthetic | SUSPENSION, EXTENDED RELEASE;SUBCUTANEOUS | 209210-001 | Oct 20, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for BYDUREON BCISE
When does loss-of-exclusivity occur for BYDUREON BCISE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09289529
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 0918904
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 34525
Estimated Expiration: ⤷ Sign Up
China
Patent: 2164597
Estimated Expiration: ⤷ Sign Up
Patent: 4248623
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0201179
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 23410
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 0299
Estimated Expiration: ⤷ Sign Up
Patent: 1170413
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Patent: 85837
Estimated Expiration: ⤷ Sign Up
Finland
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 50125
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 1231
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 44735
Estimated Expiration: ⤷ Sign Up
Patent: 51243
Estimated Expiration: ⤷ Sign Up
Patent: 12502056
Estimated Expiration: ⤷ Sign Up
Patent: 15110637
Estimated Expiration: ⤷ Sign Up
Lithuania
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 2189
Estimated Expiration: ⤷ Sign Up
Patent: 11002398
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 4997
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 41905
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 201703039S
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 41905
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1760953
Estimated Expiration: ⤷ Sign Up
Patent: 110050540
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 09178
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering BYDUREON BCISE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| South Korea | 101493102 | ⤷ Sign Up | |
| Hungary | 0300393 | C-aril glükozid SGLT2 inhibitorok és alkalmazásuk, valamint ezeket tartalmazó gyógyszerkészítmények (C-ARYL GLUCOSIDE SGLT2 INHIBITORS AND THEIR USE AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM) | ⤷ Sign Up |
| China | 1384755 | ⤷ Sign Up | |
| European Patent Office | 2016950 | Composition pharmaceutique comportant un peptide d'exendine-4 (Pharmaceutical composition comprising an exendin-4 peptide) | ⤷ Sign Up |
| Japan | 5030292 | ⤷ Sign Up | |
| Slovenia | 1283699 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BYDUREON BCISE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1734971 | 2012C/017 | Belgium | ⤷ Sign Up | PRODUCT NAME: EXENATIDE; AUTHORISATION NUMBER AND DATE: EU/1/11/696/001 20110617 |
| 1506211 | C01506211/02 | Switzerland | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN + METFORMIN; REGISTRATION NO/DATE: SWISSMEDIC 65377 16.07.2015 |
| 1506211 | SPC/GB13/021 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTERED: UK EU/1/12/795/001 20121114; UK EU/1/12/795/002 20121114; UK EU/1/12/795/003 20121114; UK EU/1/12/795/004 20121114; UK EU/1/12/795/005 20121114; UK EU/1/12/795/006 20121114; UK EU/1/12/795/007 20121114; UK EU/1/12/795/008 20121114; UK EU/1/12/795/009 20121114; UK EU/1/12/795/010 20121114 |
| 2139494 | PA2020522 | Lithuania | ⤷ Sign Up | PRODUCT NAME: SASAGLIPTINAS IR DAPAGLIFLOZINAS; REGISTRATION NO/DATE: EU/1/16/1108 20160715 |
| 1506211 | 1390017-0 | Sweden | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN OCH FARMACEUTISKT GODTAGBARA SALTER DAERAV; REG. NO/DATE: EU/1/12/795/001 20121112 |
| 1734971 | C20120006 00104 | Estonia | ⤷ Sign Up | PRODUCT NAME: EKSENATIID;REG NO/DATE: K(2011)4479 LOPLIK 23.06.2011 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


