MELINTA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for MELINTA, and when can generic versions of MELINTA drugs launch?
MELINTA has four approved drugs.
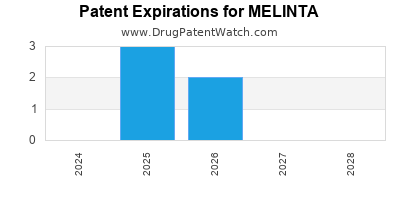
There are sixteen US patents protecting MELINTA drugs.
There are one hundred and thirty-four patent family members on MELINTA drugs in twenty-eight countries and twenty-seven supplementary protection certificates in sixteen countries.
Drugs and US Patents for MELINTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Melinta | BAXDELA | delafloxacin meglumine | TABLET;ORAL | 208610-001 | Jun 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Melinta | BAXDELA | delafloxacin meglumine | TABLET;ORAL | 208610-001 | Jun 19, 2017 | RX | Yes | Yes | RE46617 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Melinta | BAXDELA | delafloxacin meglumine | POWDER;INTRAVENOUS | 208611-001 | Jun 19, 2017 | RX | Yes | Yes | 8,410,077 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Melinta | BAXDELA | delafloxacin meglumine | POWDER;INTRAVENOUS | 208611-001 | Jun 19, 2017 | RX | Yes | Yes | 9,750,822 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Melinta | BAXDELA | delafloxacin meglumine | POWDER;INTRAVENOUS | 208611-001 | Jun 19, 2017 | RX | Yes | Yes | 7,728,143 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Melinta | BAXDELA | delafloxacin meglumine | TABLET;ORAL | 208610-001 | Jun 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Melinta | BAXDELA | delafloxacin meglumine | POWDER;INTRAVENOUS | 208611-001 | Jun 19, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MELINTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Melinta Therap | ORBACTIV | oritavancin diphosphate | POWDER;INTRAVENOUS | 206334-001 | Aug 6, 2014 | 5,840,684 | ⤷ Try a Trial |
| Melinta Therap | ORBACTIV | oritavancin diphosphate | POWDER;INTRAVENOUS | 206334-001 | Aug 6, 2014 | 5,998,581 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for MELINTA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Portugal | 3056492 | ⤷ Try a Trial |
| Hungary | S2100004 | ⤷ Try a Trial |
| Japan | 2015178515 | ⤷ Try a Trial |
| Japan | 6517725 | ⤷ Try a Trial |
| China | 107206050 | ⤷ Try a Trial |
| Japan | 2015508846 | ⤷ Try a Trial |
| Mexico | 2011002249 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for MELINTA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3214083 | 2190500-5 | Sweden | ⤷ Try a Trial | PRODUCT NAME: DELAFLOXACIN IN FREE FORM OR IN PHARMACEUTICALLY ACCEPTABLE SALT OR ESTER FORM, INCLUDING DELAFLOXACIN MEGLUMINE; REG. NO/DATE: EU/1/19/1393 20191219 |
| 3214083 | CR 2021 00002 | Denmark | ⤷ Try a Trial | PRODUCT NAME: DELAFLOXACIN ELLER ET SALT ELLER ESTER DERAF, SAERLIGT DELAFLOXACIN MEGLUMIN; REG. NO/DATE: EU/1/19/1393 20191219 |
| 2337575 | 2016019 | Norway | ⤷ Try a Trial | PRODUCT NAME: ORITAVANCIN OG FARMASOEYTISK; REG. NO/DATE: EU/1/15/989 20150415 |
| 2337575 | 2016/040 | Ireland | ⤷ Try a Trial | PRODUCT NAME: ORITAVANCIN AND PHARMACEUTICAL SALTS THEREOF, E.G DIPHOSPHATE SALTS; REGISTRATION NO/DATE: EU/1/15/989 20150319 |
| 2337575 | 122016000075 | Germany | ⤷ Try a Trial | PRODUCT NAME: ORITAVANCIN ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/15/989 20150319 |
| 3214083 | PA2021002,C3214083 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: DELAFLOKSACINAS ARBA JO DRUSKA AR ESTERIS, ISKAITANT DELAFLOKSACINO MEGLUMINA; REGISTRATION NO/DATE: EU/1/19/1393 20191216 |
| 3214083 | PA2021002 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: DELAFLOKSACINAS ARBA JO DRUSKA AR ESTERIS, ISKAITANT DELAFLOKSACINO MEGLUMINA; REGISTRATION NO/DATE: EU/1/19/1393 20191216 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.