AYTU Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AYTU, and what generic alternatives to AYTU drugs are available?
AYTU has four approved drugs.
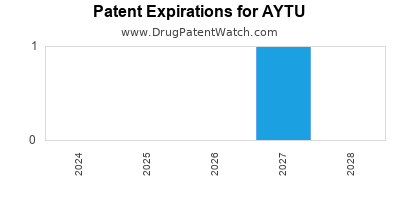
There are three US patents protecting AYTU drugs.
There are one hundred and five patent family members on AYTU drugs in sixteen countries and seven supplementary protection certificates in six countries.
Drugs and US Patents for AYTU
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aytu | ZOLPIMIST | zolpidem tartrate | SPRAY, METERED;ORAL | 022196-001 | Dec 19, 2008 | DISCN | Yes | No | 8,236,285 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Aytu Biopharma | METADATE CD | methylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 021259-004 | Feb 19, 2006 | AB2 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Aytu Biopharma | METADATE CD | methylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 021259-006 | Feb 19, 2006 | AB2 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for AYTU
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Aytu Biopharma | METADATE CD | methylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 021259-001 | Apr 3, 2001 | 6,344,215 | ⤷ Try a Trial |
| Aytu | ZOLPIMIST | zolpidem tartrate | SPRAY, METERED;ORAL | 022196-001 | Dec 19, 2008 | 7,632,517 | ⤷ Try a Trial |
| Aytu | ACIPHEX SPRINKLE | rabeprazole sodium | CAPSULE, DELAYED RELEASE;ORAL | 204736-001 | Mar 26, 2013 | 5,045,552*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for AYTU drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsules | 10 mg, 20 mg and 30 mg | ➤ Subscribe | 2005-05-13 |
| ➤ Subscribe | Extended-release Capsules | 40 mg | ➤ Subscribe | 2007-03-15 |
Premature patent expirations for AYTU
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for AYTU Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2006502148 | ⤷ Try a Trial |
| Australia | 2003270016 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2004019910 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for AYTU Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2236132 | C300714 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ZOLPIDEM EN FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN; NAT. REGISTRATION NO/DATE: RVG 108438 - 439 20130624; FIRST REGISTRATION: BE424286BE424295 2012180718 |
| 0268956 | 1999C0030 | Belgium | ⤷ Try a Trial | PRODUCT NAME: RABEPRAZOLE SODIUM; NAT. REGISTRATION NO/DATE: 5532 IE 1 F 3 19990201; FIRST REGISTRATION: GB 10555/0010 19980508 |
| 2236132 | 92636 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ZOLPIDEM ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.