Last updated: April 25, 2026
SYNTHROID (levothyroxine sodium) has sustained market leadership in U.S. thyroid-replacement therapy through broad clinical adoption, entrenched prescribing habits, and supply-chain scale. The financial trajectory is dominated by (1) long-tail demand tied to chronic hypothyroidism, (2) ongoing generic and authorized-generic competition dynamics, and (3) payer pressure that compresses unit prices while volumes remain supported.
What drives demand for SYNTHROID?
Core indication economics: chronic, not episodic
SYNTHROID is used for hypothyroidism and other thyroid hormone replacement indications that are typically lifelong. That demand profile creates:
- High repeat-purchase behavior
- Less cyclicality than acute-care drug classes
- A baseline of demand that persists despite product-level margin compression from generics
Prescriber behavior and product switching friction
Levothyroxine is therapeutically substitutable at the API level, but market access and switching remain shaped by:
- Labeling and formulation differences among products
- Clinical practice patterns that favor stable therapy
- Patient-level stability considerations that reduce rapid switching
Net effect: even when generic penetration rises, brand volumes typically decline with a lag rather than collapse.
Supply, manufacturing scale, and continuity risk
Levothyroxine demand is steady, which makes continuity of supply a competitive differentiator. Large-volume manufacturers with stable production can defend contracted share better through:
- Consistent market availability
- Pharmacy distribution reach
- Managed inventory strategies that reduce stock-outs
How does competitive pressure shape pricing and share?
Generic competition is structural, not event-based
The principal competitive dynamic for SYNTHROID is ongoing generic competition. As generic penetration increases across payers and channels, it drives:
- Lower net realized prices for the brand
- Faster erosion of pharmacy-level share where formularies change
- Shifts toward lower-cost alternatives within therapeutic-equivalent substitution frameworks
Authorized generics and channel contracting
In thyroid replacement, channel dynamics frequently include:
- Authorized generic supply where applicable, which caps brand price leverage
- PBM and payer contracting that favors “lowest cost” solutions under therapeutic-equivalence constructs
Brand vs. generics: the practical leverage is patient stability and prescriber comfort
Because levothyroxine is not a high-innovation category, competitive differentiation tends to be operational and behavioral rather than clinical novelty:
- Clinician preference for “stay on brand” in stable patients
- Patient anxiety or instability concerns after switching products
- Payer rules that may permit brand coverage with prior authorization or step edits
How has the financial trajectory typically evolved for SYNTHROID?
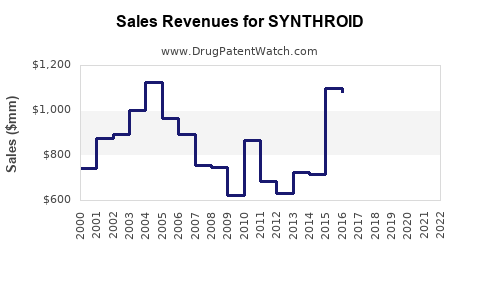
SYNTHROID’s financial trajectory over the last decade is characterized by:
- Volume resistance: chronic use keeps declines gradual versus acute drugs.
- Margin compression: price reductions and higher payer scrutiny reduce net sales per unit.
- Share drift to generics: brand share trends down as formularies and pharmacy networks broaden generic substitution.
Trajectory pattern in financial reporting (high level)
Without relying on one quarter’s noise, the prevailing pattern for mature, widely substituted brands like SYNTHROID is:
- Net sales growth is mostly absent or limited to inflation or mix effects.
- Net sales declines track generic penetration and formulary pressure.
- Operating margins compress if brand pricing falls faster than cost can scale.
What market segments matter most to SYNTHROID’s sales?
U.S. retail pharmacy and maintenance scripts
Most prescriptions are maintenance therapies, with:
- Strong representation in Medicare and commercial formularies
- High share of long-duration patients returning for refills
Institutional and specialty channels
Institutions use a mix of:
- Formulary-preferred products
- Substitution policies driven by pharmacy and contracting
While thyroid is not “specialty,” institutional formularies can still accelerate generic uptake if brand access is restricted.
Patient mix by age
Hypothyroidism prevalence rises with age. That increases structural demand support even as per-unit pricing erodes.
Where does payer policy pressure show up?
Formulary tiering and step edits
Payer management typically manifests through:
- Lower tiers or preferred generic placement
- Prior authorization requirements for brand coverage
- Step therapy rules when substitution is clinically acceptable
Pharmacy benefit design
PBMs that emphasize generic penetration tend to:
- Promote auto-substitution at point of dispensing
- Use contracting that lowers brand rebates but increases generic uptake
Net effect: persistent pricing headwinds even when volume holds.
What supply and manufacturing dynamics influence the business?
Capacity and drug availability
Levothyroxine is widely manufactured, but quality and regulatory compliance drive long-term capacity viability. For brands, supply continuity matters because:
- Stock-outs can cause longer-lasting prescriber switching to competitors
- Stable supply can preserve residual brand loyalty in the face of generics
Cost structure stability
For mature brands, cost of goods and logistics are typically less volatile than R&D-driven categories. Financial performance therefore becomes more sensitive to:
- Net pricing changes (rebates, discounts, payer mix)
- Manufacturing scale economies
- Regulatory and quality costs
What is the likely risk profile for SYNTHROID’s market position?
Category risks (generic substitution, price compression)
- Higher generic penetration reduces brand share and price.
- Authorized-generic availability can limit brand pricing recovery even if scripts remain stable.
Regulatory and quality execution
- Quality lapses or manufacturing disruptions would directly impact availability and prescribing continuity.
- Regulatory enforcement risk for any product line can affect market access.
Litigation and exclusivity dynamics (if applicable)
For mature products, brand economics are often influenced more by legal outcomes around formulation, patents, or specific product rights than by new chemical entity exclusivity. When exclusivity erodes, market outcomes shift quickly toward generics.
What should investors and R&D strategists watch next?
- Net sales per unit and rebate pressure: If declines accelerate while volumes hold, payer pressure is likely worsening.
- Share loss speed: Watch for step-function drops that indicate formulary tightening or channel switching.
- Channel availability indicators: Stock-outs at competitors can temporarily stabilize brand share, then reverse.
- Dose-level mix: Levothyroxine demand shifts across dose strengths, which can affect brand economics through pricing and substitution patterns.
- Geographic and policy shifts: Medicare Part D formulary management and PBM policy changes can swing realized pricing.
Selected product and regulatory anchor points
SYNTHROID is levothyroxine sodium. It is indicated for hypothyroidism and is also used for suppression of TSH in certain thyroid disorders and as adjunct therapy in specific clinical contexts. Prescribing and reimbursement patterns for levothyroxine reflect the broad substitution environment and chronic-use demand. (Reference: FDA product labeling and prescribing information for SYNTHROID.) [1]
Key Takeaways
- SYNTHROID’s demand is structurally supported by lifelong thyroid replacement needs, which prevents sharp volume collapse.
- Financial performance is primarily shaped by generic and authorized-generic competition that compresses net realized prices and margins over time.
- Market share erosion tends to be gradual but persistent, driven by payer formulary behavior and point-of-dispensing substitution rules.
- The biggest near- and mid-term business levers are net price (rebates and payer contracting), share drift speed, and channel continuity of supply.
FAQs
1) Is SYNTHROID primarily protected by exclusivity today?
No. In a mature levothyroxine category, brand economics typically rely on residual demand, prescriber behavior, and payer access rather than durable chemical exclusivity.
2) What matters more for SYNTHROID revenue: volume or price?
Price and net realized revenue dominate the financial trajectory because generic substitution primarily compresses brand pricing through formulary tiering, rebates, and authorized-generic contracting.
3) Do payer policies significantly affect SYNTHROID outcomes?
Yes. Formulary tiering, prior authorization, and PBM substitution design are the main mechanisms that accelerate brand-to-generic migration and reduce net pricing.
4) Does clinical switching slow generic uptake?
Yes. Stable-patient continuity and prescriber preference for maintaining consistent levothyroxine therapy can slow switching, leaving brand declines more gradual than many acute-care drugs.
5) What operational factor can temporarily change SYNTHROID share?
Supply continuity. If competitors face availability issues, brand share can temporarily stabilize or improve; once supply normalizes, substitution pressures typically reassert.
References
[1] U.S. Food and Drug Administration. (n.d.). SYNTHROID (levothyroxine sodium) prescribing information. FDA. https://www.accessdata.fda.gov