Last updated: February 19, 2026
Lamisil, a topical and oral antifungal medication whose active pharmaceutical ingredient is terbinafine hydrochloride, has maintained a significant market presence since its initial development and patenting. The drug targets dermatophytes, yeasts, and other fungi responsible for a range of superficial mycoses, including onychomycosis (nail fungus) and tinea infections (ringworm, athlete's foot, jock itch).
What is the Market Landscape for Lamisil?
The global market for antifungal drugs is substantial, driven by the high prevalence of fungal infections, particularly in aging populations and immunocompromised individuals. Lamisil, as a well-established and efficacious treatment, occupies a specific segment within this broader market.
Key Market Segments and Applications
Lamisil's primary applications fall into two main categories: topical and oral formulations.
- Topical Lamisil: This segment includes creams, gels, sprays, and solutions for the treatment of skin fungal infections like tinea pedis (athlete's foot), tinea cruris (jock itch), and tinea corporis (ringworm). Its efficacy and convenience make it a first-line treatment option for many patients.
- Oral Lamisil: The oral formulation is primarily used for onychomycosis, a more persistent and difficult-to-treat infection of the fingernails and toenails. The oral route offers systemic drug delivery, reaching the site of infection within the nail bed.
Competitive Environment
The antifungal market is characterized by both branded and generic competition.
- Branded Lamisil: Originally developed and marketed by Novartis, branded Lamisil benefits from established brand recognition and a history of clinical trials demonstrating its safety and efficacy. However, patent expiries have led to significant generic penetration.
- Generic Terbinafine Hydrochloride Products: Numerous generic manufacturers produce both topical and oral terbinafine hydrochloride formulations. These generics offer a lower price point, significantly impacting the pricing dynamics and market share of the branded product.
- Alternative Antifungal Therapies: Lamisil competes with other antifungal drug classes, including azoles (e.g., fluconazole, itraconazole) and other allylamines. The choice of therapy often depends on the type and severity of the fungal infection, patient comorbidities, and physician preference.
Market Drivers and Restraints
Several factors influence the market for Lamisil and its generic counterparts.
-
Market Drivers:
- Increasing Prevalence of Fungal Infections: Factors such as aging populations, increased use of immunosuppressive therapies, and lifestyle changes contributing to moisture retention in skin folds drive the demand for antifungal treatments.
- Growth in Onychomycosis Treatment: Onychomycosis affects an estimated 10-20% of the world's population, creating a consistent demand for effective treatments like oral terbinafine.
- Patient Preference for Topical Treatments: For superficial skin infections, topical formulations like Lamisil cream are often preferred due to their targeted action and lower systemic side effect profile.
- Generic Accessibility: The availability of affordable generic terbinafine has expanded access to treatment, increasing overall market volume.
-
Market Restraints:
- Generic Competition and Price Erosion: The presence of numerous generic manufacturers leads to intense price competition, reducing profit margins for all players.
- Potential for Drug Resistance: As with any antimicrobial agent, there is a risk of developing resistance to terbinafine, although this is less common with topical applications.
- Side Effects of Oral Terbinafine: Oral terbinafine is associated with potential side effects, including hepatotoxicity, which can limit its use in certain patient populations and requires careful monitoring. This leads to the exploration of alternative treatments.
- Long Treatment Durations for Onychomycosis: Oral terbinafine therapy for onychomycosis can require up to 12 weeks of treatment, which can affect patient adherence.
What is the Patent Landscape and Exclusivity Status of Lamisil?
The intellectual property surrounding Lamisil has been critical to its commercial success and subsequent market evolution.
Key Patents and Their Lifespans
-
Original Compound Patents: The core patent for terbinafine hydrochloride was filed by Sandoz (later acquired by Novartis) in the early 1980s.
- US Patent 4,355,031: This patent covers the terbinafine compound itself and its therapeutic use for treating fungal infections. It was filed on December 30, 1976, and granted on October 18, 1982. Its expiration allowed for the development of generic versions.
- European Patent EP0040349B1: This European patent provided protection in key European markets.
-
Formulation and Method of Use Patents: Over time, additional patents were sought for specific formulations (e.g., stable topical compositions, specific oral dosage forms) and novel methods of treatment. However, the primary exclusivity was tied to the compound patent.
Patent Expiry and Generic Entry
The expiration of the foundational compound patents for terbinafine hydrochloride was the primary catalyst for generic entry.
- United States: The primary compound patent in the US expired in the early 2000s. This allowed generic manufacturers to launch their versions of terbinafine hydrochloride.
- European Union: Similar patent expiries occurred across major European countries, leading to widespread generic availability.
Current Exclusivity and Market Position
As of the current date, there is no active patent exclusivity for the original terbinafine hydrochloride compound itself.
- Branded Dominance Shift: Novartis's branded Lamisil has transitioned from a period of market exclusivity to competing with a highly fragmented generic market.
- Generic Market Share: The vast majority of the current market for terbinafine hydrochloride is served by generic products.
- Potential for New IP: While the core compound is off-patent, companies may still pursue patents related to novel delivery systems, combination therapies, or specific indications that could create limited periods of new exclusivity, although such developments have not significantly reshaped the broad market for terbinafine hydrochloride.
What is the Financial Trajectory of Lamisil?
The financial trajectory of Lamisil reflects its lifecycle from a blockbuster branded drug to a widely available generic medication.
Peak Sales and Revenue Generation
During its period of patent exclusivity, branded Lamisil generated substantial revenue for Novartis.
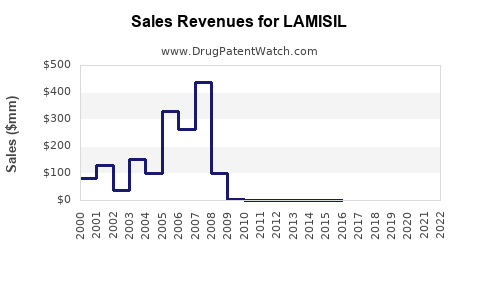
- Peak Sales: While precise peak annual sales figures are proprietary, reports indicate that branded Lamisil (including both topical and oral formulations) achieved peak annual sales exceeding $1 billion USD globally in the early 2000s. This revenue was driven by its strong efficacy, particularly for onychomycosis, where it represented a significant therapeutic advance.
Post-Patent Expiry Revenue Decline
Upon the expiration of its primary patents, the revenue trajectory for branded Lamisil experienced a significant decline due to the influx of generic competition.
- Revenue Erosion: The introduction of generics led to rapid price reductions. Branded Lamisil's market share diminished as physicians and patients opted for lower-cost generic alternatives.
- Current Revenue for Branded Product: While still marketed, the revenue generated by branded Lamisil is a fraction of its peak, primarily serving a niche of consumers who prefer the brand or have specific reasons for avoiding generics.
Generic Terbinafine Market Growth
The financial impact has shifted from branded sales to the collective revenue generated by the generic terbinafine hydrochloride market.
- Market Size: The global market for generic terbinafine hydrochloride is estimated to be in the hundreds of millions of dollars annually, driven by volume rather than high per-unit pricing. This market is fragmented among numerous manufacturers.
- Profitability Dynamics: For generic manufacturers, profitability is achieved through high-volume production and efficient supply chains, often with narrower profit margins per unit compared to the peak profitability of the branded drug.
- Key Players in Generics: Companies such as Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and numerous smaller regional manufacturers are significant players in the generic terbinafine market.
Pricing Trends
Pricing trends for terbinafine hydrochloride have moved from premium branded pricing to highly competitive generic pricing.
- Branded Pricing: At its peak, branded Lamisil commanded premium pricing reflecting its R&D investment and market exclusivity.
- Generic Pricing: Generic terbinafine hydrochloride products are priced at a significant discount, often 60-80% lower than the peak branded price, depending on the region, dosage form, and volume.
- Price Stability vs. Volatility: While branded pricing was relatively stable during exclusivity, generic pricing can exhibit some volatility due to supply and demand dynamics, competitive pressures, and the purchasing power of large distributors and pharmacy benefit managers.
Investment Outlook
Investment in Lamisil as a branded product is minimal. The investment focus has shifted to the generic manufacturing and distribution of terbinafine hydrochloride.
- Generic Manufacturing Investment: Companies investing in this space focus on optimizing manufacturing processes for cost efficiency, ensuring robust supply chains, and navigating regulatory requirements in different markets.
- R&D Investment: New R&D investment related to terbinafine hydrochloride is unlikely to focus on the compound itself but could explore novel drug delivery systems or fixed-dose combinations if a compelling clinical rationale emerges. However, the established efficacy and low cost of generics limit the economic incentive for significant new R&D.
Key Takeaways
- Lamisil (terbinafine hydrochloride) has transitioned from a high-revenue branded antifungal to a widely available generic medication following patent expiries.
- The global market for terbinafine hydrochloride is now dominated by generic products, with significant price erosion from its branded peak sales exceeding $1 billion annually.
- The primary drivers for the continued market demand are the high prevalence of fungal infections, particularly onychomycosis, and the accessibility of affordable generic treatments.
- While branded Lamisil's financial peak has passed, the generic market for terbinafine hydrochloride remains substantial, serving hundreds of millions of dollars in annual revenue driven by volume.
- Investment in the terbinafine hydrochloride space is focused on cost-efficient generic manufacturing and distribution rather than novel R&D for the parent compound.
FAQs
- What is the current patent status of Lamisil (terbinafine hydrochloride)?
The original compound patents for terbinafine hydrochloride have expired globally, allowing for the widespread production and sale of generic versions.
- How has the financial performance of Lamisil changed since its patent expiry?
Branded Lamisil's revenue significantly declined from its peak exceeding $1 billion annually following generic entry. The financial impact has shifted to the collective revenue of the generic terbinafine hydrochloride market.
- Who are the main competitors to Lamisil?
Lamisil faces competition from a broad range of generic terbinafine hydrochloride products and other classes of antifungal drugs, such as azoles (e.g., fluconazole, itraconazole).
- What are the primary indications for Lamisil?
Lamisil is indicated for the treatment of fungal infections, including onychomycosis (nail fungus), tinea pedis (athlete's foot), tinea cruris (jock itch), and tinea corporis (ringworm).
- What is the investment outlook for Lamisil?
Investment opportunities are primarily in the generic manufacturing and distribution of terbinafine hydrochloride, focusing on cost-efficiency and supply chain management, rather than on the development of new branded Lamisil products.
Citations
[1] U.S. Patent 4,355,031. (1982). Therapeutic agents and method for treating fungal infections. Sandoz, Ltd.
[2] European Patent EP0040349B1. (1988). Process for the preparation of N-(6,6-dimethyl-2-hepten-4-ynyl)-N-methyl-3-phenyl-2-propenylamine and salts thereof. Sandoz, A.G.
[3] IMS Health. (Data accessed for market reports covering antifungal drug sales and market share).
[4] Various pharmaceutical industry news and market research reports on antifungal drug markets and generic drug penetration.