CELLCEPT Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cellcept, and when can generic versions of Cellcept launch?
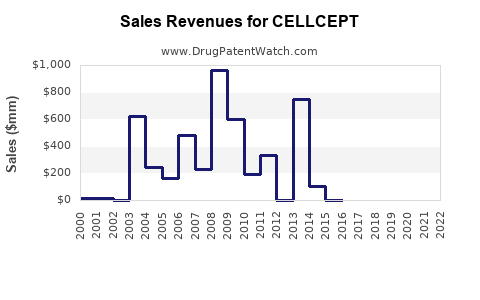
Cellcept is a drug marketed by Roche Palo and is included in four NDAs.
The generic ingredient in CELLCEPT is mycophenolate mofetil hydrochloride. There are thirty-eight drug master file entries for this compound. Twelve suppliers are listed for this compound. Additional details are available on the mycophenolate mofetil hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Cellcept
A generic version of CELLCEPT was approved as mycophenolate mofetil hydrochloride by PAR STERILE PRODUCTS on October 28th, 2016.
Summary for CELLCEPT
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 4 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 152 |
| Clinical Trials: | 383 |
| Patent Applications: | 4,341 |
| Formulation / Manufacturing: | see details |
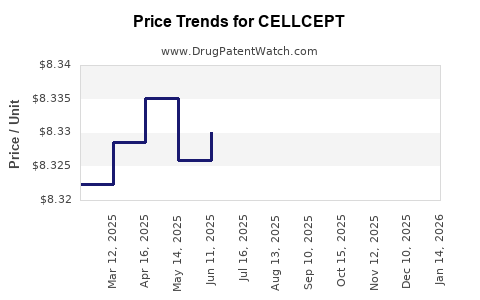
| Drug Prices: | Drug price information for CELLCEPT |
| What excipients (inactive ingredients) are in CELLCEPT? | CELLCEPT excipients list |
| DailyMed Link: | CELLCEPT at DailyMed |
Recent Clinical Trials for CELLCEPT
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of California, Davis | Phase 3 |
| Medical College of Wisconsin | Phase 2 |
| Paladin Labs Inc. | Phase 4 |
Pharmacology for CELLCEPT
| Drug Class | Antimetabolite Immunosuppressant |
Paragraph IV (Patent) Challenges for CELLCEPT
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CELLCEPT | For Oral Suspension | mycophenolate mofetil | 200 mg/mL | 050759 | 1 | 2011-03-25 |
US Patents and Regulatory Information for CELLCEPT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Roche Palo | CELLCEPT | mycophenolate mofetil | CAPSULE;ORAL | 050722-001 | May 3, 1995 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Roche Palo | CELLCEPT | mycophenolate mofetil | TABLET;ORAL | 050723-001 | Jun 19, 1997 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Roche Palo | CELLCEPT | mycophenolate mofetil | FOR SUSPENSION;ORAL | 050759-001 | Oct 1, 1998 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Roche Palo | CELLCEPT | mycophenolate mofetil hydrochloride | INJECTABLE;INJECTION | 050758-001 | Aug 12, 1998 | AP | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CELLCEPT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Roche Palo | CELLCEPT | mycophenolate mofetil | FOR SUSPENSION;ORAL | 050759-001 | Oct 1, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Roche Palo | CELLCEPT | mycophenolate mofetil hydrochloride | INJECTABLE;INJECTION | 050758-001 | Aug 12, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Roche Palo | CELLCEPT | mycophenolate mofetil | CAPSULE;ORAL | 050722-001 | May 3, 1995 | ⤷ Try a Trial | ⤷ Try a Trial |
| Roche Palo | CELLCEPT | mycophenolate mofetil | TABLET;ORAL | 050723-001 | Jun 19, 1997 | ⤷ Try a Trial | ⤷ Try a Trial |
| Roche Palo | CELLCEPT | mycophenolate mofetil hydrochloride | INJECTABLE;INJECTION | 050758-001 | Aug 12, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for CELLCEPT
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Passauer Pharma GmbH | Myclausen | mycophenolate mofetil | EMEA/H/C/001218 Myclausen is indicated in combination with ciclosporin and corticosteroids for the prophylaxis of acute transplant rejection in patients receiving allogeneic renal, cardiac or hepatic transplants., |
Authorised | yes | no | no | 2010-10-07 | |
| Roche Registration GmbH | CellCept | mycophenolate mofetil | EMEA/H/C/000082 CellCept is indicated in combination with ciclosporin and corticosteroids for the prophylaxis of acute transplant rejection in patients receiving allogeneic renal, cardiac or hepatic transplants. |
Authorised | no | no | no | 1996-02-14 | |
| Teva B.V. | Myfenax | mycophenolate mofetil | EMEA/H/C/000884 Myfenax is indicated in combination with ciclosporin and corticosteroids for the prophylaxis of acute transplant rejection in patients receiving allogeneic renal, cardiac or hepatic transplants. |
Authorised | yes | no | no | 2008-02-21 | |
| Teva Pharma B.V. | Mycophenolate mofetil Teva | mycophenolate mofetil | EMEA/H/C/000882 Mycophenolate mofetil Teva is indicated in combination with ciclosporin and corticosteroids for the prophylaxis of acute transplant rejection in patients receiving allogeneic renal, cardiac or hepatic transplants. |
Authorised | yes | no | no | 2008-02-21 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for CELLCEPT
See the table below for patents covering CELLCEPT around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 0722294 | CONNECTEUR AMELIORE PERMETTANT DE COUPLER UN TRANSDUCTEUR A ULTRASONS A UN CATHETER A ULTRASONS (IMPROVED CONNECTOR FOR COUPLING AN ULTRASOUND TRANSDUCER TO AN ULTRASOUND CATHETER) | ⤷ Try a Trial |
| Canada | 2172506 | SUSPENSIONS DE MYCOPHENOLATE MOFETILE A DOSAGE ELEVE ET USAGE ORAL (MYCOPHENOLATE MOFETIL AND MYCOPHENOLIC ACID HIGH DOSE ORAL SUSPENSIONS) | ⤷ Try a Trial |
| Finland | 20060003 | ⤷ Try a Trial | |
| Luxembourg | 88795 | ⤷ Try a Trial | |
| Romania | 115412 | COMPOZITIE FARMACEUTICA SI PROCEDEU DE OBTINERE A COMPOZITIEI FARMACEUTICE (PHARMACEUTICAL COMPOSITION AND PROCESS FOR PRODUCING THE SAME) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CELLCEPT
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0281713 | SPC/GB96/026 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MYCOPHENOLATE MOFETIL OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: CH 53337 19951103; CH 53338 19951103; UK EU/1/96/005/001 19960214; UK EU/96/005/002 19960214 |
| 0281713 | 96C0031 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MYCOPHENOLATE MOFETIL; NAT. REGISTRATION NO/DATE: EU/1/96/005/001 19960214; FIRST REGISTRATION: CH 53337 19951103 |
| 0281713 | C960019 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: MYCOFENOLAAT MOFETIL, DESGEWENST IN DE VORM VAN EEN FARMACEUTIS CH AANVAARDBAAR ZOUT, OF IN DE VORM VAN EEN ESTER MET EEN CARBO NZUUR MET DE FORMULE RCOOH WAARIN R EEN ALKYL- OF CYCLOALKYLGRO EP MET 1-6 KOOLSTOFATOMEN IS OF EEN UIT AL DAN NIET GESUBSTITUE; REGISTRATION NO/DATE: EU/1/96/005/001 - EU/1/96/005/002 19960214, IKS 53337-01 19951103 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


