Last updated: February 19, 2026
Methylphenidate, a central nervous system stimulant primarily used for treating Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy, exhibits a mature yet consistently growing market. Its efficacy in managing core ADHD symptoms, coupled with an expanding diagnostic landscape and the availability of diverse formulations, underpins sustained demand. Intellectual property remains a significant factor, with patent expiries opening avenues for generic competition and influencing pricing strategies, while ongoing research into novel delivery systems and broader therapeutic applications continues to shape future market potential.
What are the Key Market Drivers for Methylphenidate?
Several factors drive the methylphenidate market. The increasing prevalence of ADHD diagnoses globally is a primary catalyst. Factors contributing to this rise include enhanced awareness among healthcare professionals and the public, improved diagnostic tools, and a greater understanding of the neurobiological underpinnings of the disorder.
- Rising ADHD Diagnoses: Global ADHD diagnoses have shown a consistent upward trend. In the United States, for instance, estimates suggest that approximately 11% of children aged 4-17 have received an ADHD diagnosis at some point [1]. This diagnostic growth translates directly into increased demand for methylphenidate.
- Expanded Age Indications: While initially approved for pediatric use, methylphenidate's efficacy has been recognized and approved for adolescent and adult populations. This broadening of approved age groups significantly expands the patient pool seeking treatment.
- Formulation Diversity: The market offers a wide array of methylphenidate formulations, catering to different patient needs and preferences. These include immediate-release (IR), extended-release (ER), and transdermal patch delivery systems. This diversity enhances patient adherence and treatment effectiveness.
- Immediate-Release (IR): Provides rapid symptom control but requires multiple daily doses.
- Extended-Release (ER): Offers sustained symptom management with once-daily dosing, improving convenience and adherence. Examples include Concerta (Ortho-McNeil Pharmaceutical) and Ritalin LA (Novartis).
- Transdermal Patch: Provides a steady release of medication over a defined period, offering an alternative for patients who have difficulty swallowing pills or require precise dosage titration. Daytrana (Novartis) is an example.
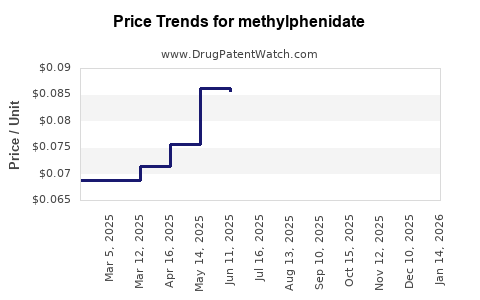
- Generic Competition: The expiry of key patents for branded methylphenidate products has led to the proliferation of generic alternatives. This competition has generally lowered prices, making the medication more accessible and increasing overall volume consumption.
What is the Patent Landscape for Methylphenidate?
The patent landscape for methylphenidate is characterized by the expiration of primary composition of matter patents, allowing for widespread generic entry. However, innovation continues through patents protecting novel formulations, delivery systems, and manufacturing processes.
| Patent Holder/Developer |
Original Drug Product |
Key Composition of Matter Patent Expiry (Approximate) |
Current Status |
| Ciba-Geigy (now Novartis) |
Ritalin (IR) |
Early 2000s |
Generic availability widespread. |
| Johnson & Johnson |
Concerta (ER) |
Mid-2010s |
Generic versions of OROS delivery system available from multiple manufacturers. |
| Noven Pharmaceuticals |
Daytrana (Transdermal) |
Early 2020s |
Patent landscape complex, with ongoing litigation and generic development. |
| Multiple Generic |
Various generic formulations |
N/A (post-expiry) |
Significant market share secured through lower pricing. |
The expiration of the core patent for the original Ritalin formulation in the early 2000s marked a significant shift, enabling generic manufacturers to enter the market. This was followed by the patent expiry of Concerta's proprietary OROS (Osmotic Release Oral System) technology, further increasing generic penetration. While the original patents have largely expired, companies continue to seek patent protection for innovative delivery methods, such as advanced extended-release mechanisms or novel transdermal systems, and for specific therapeutic uses or combinations. These secondary patents can create new market exclusivity periods for specific branded products.
How Has the Financial Trajectory of Methylphenidate Evolved?
The financial trajectory of methylphenidate has transitioned from a high-growth phase driven by branded products to a more stable, volume-driven market influenced by generic competition.
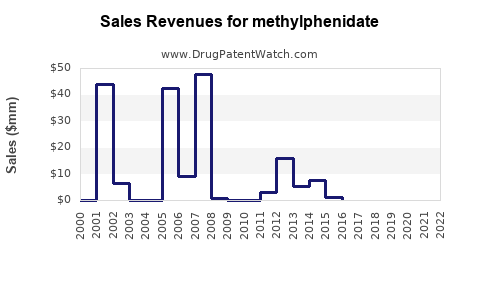
- Branded Product Revenue: Historically, branded methylphenidate products like Ritalin and Concerta generated substantial revenues. For example, Novartis’s Ritalin and Concerta sales were significant contributors to its pharmaceutical division. In 2007, Concerta alone reported global sales of $1.4 billion [2].
- Impact of Generic Entry: The entry of generic methylphenidate has led to a significant decline in the average selling price (ASP) for the drug class. While the overall market volume has increased, the revenue generated per unit has decreased. This shift necessitates a focus on manufacturing efficiency and market share volume for generic producers.
- Market Size and Growth: The global methylphenidate market was valued at approximately $4.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4-6% through 2030, driven by increasing diagnoses and broader application [3]. This growth is more moderate compared to the peak years of branded exclusivity but signifies a sustained demand.
- Key Market Players: The market is fragmented, with major branded manufacturers like Novartis and Johnson & Johnson still holding significant positions through their established brands, alongside a robust segment of generic pharmaceutical companies. Key generic players include Teva Pharmaceuticals, Mylan (now Viatris), and Perrigo.
The financial trajectory demonstrates a typical lifecycle for a successful pharmaceutical product. Initial exclusivity and innovation command premium pricing and high revenue growth. Upon patent expiry, generic competition erodes pricing power, leading to a market characterized by volume sales and cost-efficient manufacturing. The continued moderate growth indicates that the underlying demand for ADHD treatment remains strong, even with price pressures.
What are the Key Challenges and Opportunities in the Methylphenidate Market?
The methylphenidate market faces several challenges, primarily related to regulatory scrutiny and the emergence of alternative treatments. However, opportunities exist in product innovation and expanded therapeutic applications.
Challenges:
- Regulatory Scrutiny and Diversion Concerns: Methylphenidate is a Schedule II controlled substance in the United States due to its potential for abuse and diversion. This classification imposes strict regulations on manufacturing, distribution, and prescribing, creating operational hurdles and compliance costs.
- Side Effect Profile and Patient Adherence: While generally well-tolerated, methylphenidate can cause side effects such as insomnia, decreased appetite, and cardiovascular issues. These can impact patient adherence to treatment, leading to suboptimal outcomes.
- Competition from Alternative ADHD Treatments: The ADHD treatment landscape is evolving. Non-stimulant medications, behavioral therapies, and emerging non-pharmacological interventions present alternative or complementary treatment options that can reduce reliance on methylphenidate for some patient populations.
- Pricing Pressures from Payers: Given the widespread availability of generics and the established nature of the drug, payers often exert significant pressure on pricing, further impacting the profitability of both branded and generic manufacturers.
Opportunities:
- Development of Abuse-Deterrent Formulations: Research into and development of abuse-deterrent formulations (ADFs) for methylphenidate can address diversion concerns and potentially improve regulatory standing, creating a competitive advantage.
- Novel Drug Delivery Systems: Continued innovation in drug delivery systems, such as longer-acting formulations or alternative routes of administration, can enhance patient convenience, improve adherence, and potentially command premium pricing for differentiated products.
- Expanded Therapeutic Indications: Ongoing research may identify new therapeutic uses for methylphenidate beyond ADHD and narcolepsy. If successful, this could open entirely new market segments.
- Growth in Emerging Markets: As awareness and diagnostic capabilities improve in emerging economies, the demand for ADHD treatments, including methylphenidate, is expected to rise, presenting significant growth potential.
What is the Future Outlook for Methylphenidate?
The future outlook for methylphenidate remains positive, characterized by continued demand driven by the persistent prevalence of ADHD and its established therapeutic profile. The market will likely see a bifurcation: stable, high-volume generic sales and targeted innovation in specialty formulations or new indications.
The increasing recognition of ADHD in adults and the ongoing global expansion of diagnostic services will continue to fuel demand. Generic manufacturers will likely focus on optimizing production costs and securing market share through competitive pricing. For branded pharmaceutical companies, the focus will be on developing differentiated products. This could involve novel extended-release technologies that offer superior patient convenience or extended duration of action, or formulations that mitigate specific side effects.
Furthermore, potential exploration of methylphenidate for off-label uses or as an adjunct in other neurological or psychiatric conditions, if supported by robust clinical evidence, could unlock new revenue streams. However, the drug’s status as a controlled substance will necessitate ongoing stringent regulatory compliance and a careful approach to market expansion. The competitive landscape will remain dynamic, with a constant interplay between cost-effective generic supply and value-added branded innovations.
Key Takeaways
- The methylphenidate market is driven by increasing ADHD diagnoses across pediatric, adolescent, and adult populations, coupled with a wide range of available formulations.
- Primary composition of matter patents for established methylphenidate products have expired, leading to significant generic competition and lower average selling prices.
- While branded product revenues have historically been high, the market now relies on volume, with a projected CAGR of 4-6% through 2030, valued at approximately $4.5 billion in 2022.
- Key challenges include regulatory controls due to its Schedule II status, potential side effects impacting adherence, and competition from alternative ADHD treatments.
- Opportunities lie in developing abuse-deterrent formulations, novel drug delivery systems, exploring expanded therapeutic indications, and capitalizing on growth in emerging markets.
- The future outlook indicates sustained demand, with generic markets focusing on volume and branded sectors prioritizing innovation in delivery systems and potential new uses.
Frequently Asked Questions
-
What is the primary difference between immediate-release and extended-release methylphenidate formulations regarding patient impact?
Immediate-release (IR) formulations of methylphenidate provide rapid symptom control but require multiple daily doses, which can be disruptive to a patient's daily routine and potentially lead to inconsistent symptom management. Extended-release (ER) formulations offer sustained symptom relief with a single daily dose, improving convenience, adherence, and providing more stable therapeutic levels throughout the day.
-
How do patent expiries specifically impact the pricing of methylphenidate drugs?
When primary patents expire, it allows multiple generic pharmaceutical companies to manufacture and sell their versions of the drug. This increased competition among manufacturers of generic methylphenidate drives down the average selling price (ASP) significantly compared to the period when only the branded product was available.
-
What are the main regulatory hurdles faced by methylphenidate manufacturers?
Methylphenidate is classified as a Schedule II controlled substance in the United States and similarly regulated in other countries. This classification imposes stringent requirements for manufacturing quotas, security, record-keeping, and distribution to prevent diversion and abuse. Compliance with these regulations adds significant operational costs and complexities.
-
Besides ADHD and narcolepsy, are there any other therapeutic areas where methylphenidate is being investigated or used?
While ADHD and narcolepsy are the primary FDA-approved indications for methylphenidate, research has explored its potential use in other conditions. These include treatment-resistant depression, cognitive enhancement in certain neurological conditions, and management of fatigue in specific patient populations. However, these are largely investigational and not widely established clinical uses.
-
What role do alternative ADHD treatments play in the market share of methylphenidate?
Alternative ADHD treatments, including non-stimulant medications (such as atomoxetine, guanfacine, and clonidine), behavioral therapy, and emerging interventions, present a competitive challenge to methylphenidate. For patients who experience significant side effects from stimulants, for whom stimulants are contraindicated, or as a complementary approach, these alternatives can reduce the overall reliance on methylphenidate.
Cited Sources
[1] Centers for Disease Control and Prevention. (2023, August 1). Attention-Deficit/Hyperactivity Disorder (ADHD) in Children. U.S. Department of Health and Human Services. https://www.cdc.gov/ncbddd/adhd/data.html
[2] Novartis AG. (2008, February 13). Novartis Reports 2007 Full Year Results. [Press Release].
[3] Grand View Research. (2023). Methylphenidate Market Size, Share & Trends Analysis Report By Type (Immediate Release, Extended Release), By Application (ADHD, Narcolepsy), By Region, And Segment Forecasts, 2023 - 2030. [Market Research Report].